Angiogenesis inhibitors
Angiostatin
Naturally occurring protein found in some animal species. Angiostatin is known to be cleaved by MMPs [2]. Angiostatin binds to endothelial cell surface adenosine triphosphate (ATP) synthase and angiomotin.
Bevacizumab
A humanized monoclonal antibody. The first commercially available angiogenesis inhibitor. Inhibits the actions of VEGF by binding directly to VEGF-A. Used primarily for colorectal cancer [3]. Usually used along with combination drug chemotherapy.
Celecoxib
A nonsteroidal anti-inflammatory drug (NSAID). Highly selective COX-2 inhibitor. This selectivity helps reduce stomach ulcers.
Curcumin
Epigallocatechin gallate
A flavanoid class molecule that acts as a powerful antioxidant and protects against oxidative stress and free radical damage.
Endostatin
A C-terminal fragment derived from type 18 collagen. It is a broad-spectrum angiogenesis inhibitor that interferes with the proangiogenic action of growth factors, bFGF and VEGF.
Imatinib mesylate
Acts by inhibiting particular tyrosine kinase (TK) enzymes, instead of nonspecifically inhibiting rapidly dividing cells. It occupies the TK active site, leading to a decrease in bcr-abl transformation. Imatinib mesylate is especially useful in that it is one of the few tyrosine kinase inhibitors with appropriate selectivity and limited toxicity. Studies show that imatinib may be also useful in treating smallpox [6].
Honokiol
Melanoma therapy under development. Relevant in that IFN-α, the only widely prescribed adjuvant therapy for melanoma, has marked side effects. A biphenolic ring with an ortho-allyl moiety, honokiol induced caspase-dependent cell death in B-CLL cells [7] and a variety of melanoma cell lines. Mechanism of action is likely direct inhibition of GRP78 and activation of Sirt3 [8].
Silymarin
Silymarin consists of a family of flavonoids commonly found in the dried fruit of the milk thistle plant. Extensive research has shown that silymarin can suppress the proliferation of a variety of tumor cells. This is accomplished by inhibition of cell-survival kinases (AKT and MAPK) and inhibition of inflammatory transcription factors (NF-kβ). It can also downregulate gene products involved in the proliferation of tumor cells (COX-2), invasion (MMP-9), angiogenesis (VEGF), and metastasis [9].
Thalidomide
Thalidomide inhibits the release of TNF-α from monocytes and modulates other cytokine action. Thalidomide may act to heal aphthous ulcers by inhibiting angiogenesis and promoting reepithelialization [10]. Thalidomide is also useful in the treatment of multiple myelomas, autoimmune diseases, and leprosy [11].
Gentian violet
Solenopsin
Rapamycin
Macrocyclic triene antibiotic, also known as sirolimus, possessing immunosuppressant and antiproliferative properties. Works through inhibition of mTOR, leading to interruption of IL-2 signaling and a cell cycle arrest at G1–S [18]. In preliminary trials for treatment of angiomyolipomas and brain tumors associated with tuberous sclerosis [19]. Used in the treatment of Kaposi’s sarcoma in patients receiving renal transplants [20].
Angiogenesis stimulators
Vascular endothelial growth factor (VEGF)
The upregulation of VEGF is the main operator in the physiologic response during exercise. Muscle contraction increases the blood flow to the affected areas. This increased flow causes an increase in the mRNA production of VEGF receptors 1 and 2. This increase in receptors increases the signaling cascades related to angiogenesis.
Basic fibroblast growth
factor (bFGF)
Basic fibroblast growth factor is a member of the fibroblast growth factor family. In normal tissue, basic fibroblast growth factor is present in basement membranes and in the subendothelial extracellular matrix of blood vessels. bFGF stays membrane-bound as long as there is no signal peptide. During both wound healing of normal tissues and tumor development, the action of heparan sulfate–degrading enzymes activates bFGF, thus mediating the formation of new blood vessels (angiogenesis).
Matrix metalloproteinase (MMP)
MMPs help degrade the proteins that keep the vessel walls solid. This proteolysis allows the endothelial cells to escape into the interstitial matrix as seen in sprouting angiogenesis. These enzymes are highly regulated during the vessel formation process because this destruction of the extracellular matrix would destroy the integrity of the microvasculature. MMP2 and MMP9 are the two proteinases linked to angiogenesis.
Cyclooxygenase-2
(COX2)– prostaglandin E2 (PGE2)
COX-2 inhibitors are a class of nonsteroidal anti-inflammatory drugs (NSAIDs) that selectively block the COX-2 enzyme. This action impedes the production of the chemical messengers (prostaglandins) that cause the pain and swelling of arthritis inflammation. Being that COX-2 inhibitors selectively block the COX-2 enzyme and not the COX-1 enzyme, these drugs are uniquely different from traditional NSAIDs.
Platelet-derived
Growth factor (PDGF)
PDGF is a dimer that activates its signaling pathway by a ligand-induced receptor dimerization and autophosphorylation. PDGF has provided a market for protein receptor antagonists to treat disease. These antagonists include specific antibodies that target the molecule of interest.
Infectious Processes
Neither acute nor chronic infections have traditionally been considered angiogenic processes, but the resolution of an acute infection and the maintenance of a chronic infection require an intact angiogenic system. Colonization and invasion of the skin by both gram-positive and gram-negative organisms activates the innate immune system, which results in the production of antimicrobial peptides, some of which also impact endothelial cells. While the activation of the innate immune system is sufficient to control and eliminate many bacterial colonizations and invasions, it is not sufficient to control high inocula of infection or particularly virulent bacterial or viral infections. Therefore, danger signals are transmitted systemically, which allow for cellular reinforcements. The arrival of cellular reinforcements requires an intact vascular system, which can respond to infection by activation of endothelial adhesion molecules (vascular cell adhesion molecule [VCAM], intercellular adhesion molecule [ICAM], E-selectin), allowing neutrophils, lymphocytes, and macrophages to proceed to the site of infection with selectivity (Fig. 11.1).
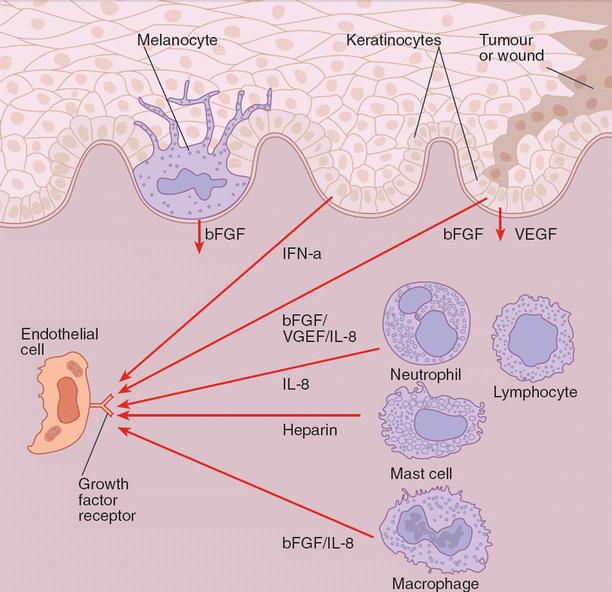

Fig. 11.1
Basement membrane-reservoir of acidic fibroblast growth factor (aFGF) and basic fibroblast growth factor (bFGF) bound to heparan sulfate proteoglycan leads to the disruption of basement membrane which leads to release of growth factor. Keratinocyte-derived interferon-α (IFN-α) directly inhibits endothelial growth. Upon activation of a growth factor receptor by a growth factor, the endothelial cell is stimulated to proliferate, produce proteases that migrate toward the source of growth factors, and form tubes, the precursors of capillaries
One of the most graphic demonstrations of the importance of this system is the genetic deficiency of CD11, a neutrophil adhesion molecule that is required for the exit of neutrophils from blood vessels. Patients lacking CD11 develop neutrophilia due to inflammatory stimuli, but these neutrophils are unable to leave the blood vessel, leading to “cold” staphylococcal abscesses and high neutrophil counts due to the inability of these neutrophils to reach the site of infection. In the absence of bone marrow transplantations, these patients succumb to staphylococcal sepsis. Acute infections like colonization of atopic dermatitis by gram-positive organisms e.g. Staphylococcus and Streptococcus cause exacerbation of Th2 type of inflammation. A major player in the vascular permeability of gram-positive infections is angiopoietin-2 (Ang2). Recently we described elevated levels of Ang2 in superinfected atopic dermatitis and demonstrated that the NADPH oxidase inhibitor, gentian violet, is useful in the treatment of atopic dermatitis, due to both downregulation of host Ang-2 and killing of gram-positive organisms [14].
Chronic infections are usually the result of bacteria capable of intracellular colonization (treponemes, mycobacterium, Bartonella), viral infections that are capable of latency, or viral oncogenes (human papillomavirus [HPV], herpes virus). Bacterial infections often colonize cells of endothelial or monocyte/macrophage origin, and in retrospect, this should not be surprising. These cells share a common precursor cell in the hemangioblast. As opposed to most extracellular bacteria, chronic bacterial infections of the skin often manifest after a systemic infection, perhaps due to infection of the hemangioblast, with preferential colonization of the infected endothelial cell or monocyte in the dermis. Of note, there are two major receptors for VEGF, the major chemotactic angiogenic factor, and these are differentially expressed in the descendants of the hemangioblasts. Endothelial cells express primarily VEGFR2, while monocytes/macrophages express VEGFR1. Blockade of each of these receptors with specific antibodies impairs angiogenesis, thus demonstrating a role of both endothelial cells and hematopoietic cells in angiogenesis. These blockades likely results in the highly impaired wound healing in patients who are neutropenic.
Recently, tissue hypoxia has been shown to be a factor favoring the persistence of intracellular organisms such as Leishmania. This may also hold true of cutaneous tuberculosis, in which tissue hypoxia may prevent access of bactericidal nitric oxide [21].
Outside of embryonic cells, angiogenesis is affected primarily by the endothelial cell in two ways: through recruitment of endothelial cells from local mesenchymal cells, and through recruitment and trans-differentiation of bone marrow-derived stem cells into endothelial cells. As cells may be derived from either of these sources, the efficacy of an antiangiogenic treatment is dependent on the source of cells in a particular lesion. For instance, in advanced malignancy, tumor cells can form vascular channels and phenotypically resemble endothelial cells; this process is known as vascular mimicry. Thus, for the development of an efficacious angiogenesis inhibitor, and for a proper understanding of the dynamic and unique nature of a lesion, the mechanisms of angiogenesis must be understood.
One of the cornerstones in the development of angiogenesis inhibitors is the angiogenic “switch.” In a given cell, there is a balance between pro- and antiangiogenic signals. As cells become more malignant, a threshold is crossed as the balance shifts toward the proangiogenic state. It follows that cells produce angiogenesis stimulators and direct or indirect inhibitors. The direct inhibitors diminish endothelial cells’ development of neovasculature. Indirect inhibitors block the angiogenic stimulatory pathway through inhibition of growth factors such as VEGF or the basic fibroblast growth factor (bFGF), or through blockade of releasing factors such as the corticotropin-releasing factor [22]. All of this functions, in the end, to impair cells’ ability to promote angiogenesis.
The regulation of angiogenic growth factors is often complex, but the best elucidated is the regulatory pathways surrounding VEGF. Several stimuli regulate VEGF; on a transcriptional level there are AP1 and Sp1, and on a translational level there are hypoxia-inducible factor 2α (HIF-2α) and mitogen-activated protein kinase (MAPK) (Fig. 11.2). Though much research has focused on inhibition of HIF-1, selective HIF-2α inhibition is emerging as a more vital protein in VEGF transcription, or the inhibition thereof. The fact that HIF1-deficient cells are capable of forming aggressive tumors implicates HIF1-independent processes in VEGF upregulation. Though many specific and selective inhibitors of HIF, akt, and a variety of other VEGF signaling molecules are under development, it must be understood that, like many biologic systems, tumors are dynamic and capable of switching signaling pathways, for example from HIF1-dependence to HIF1-independence.
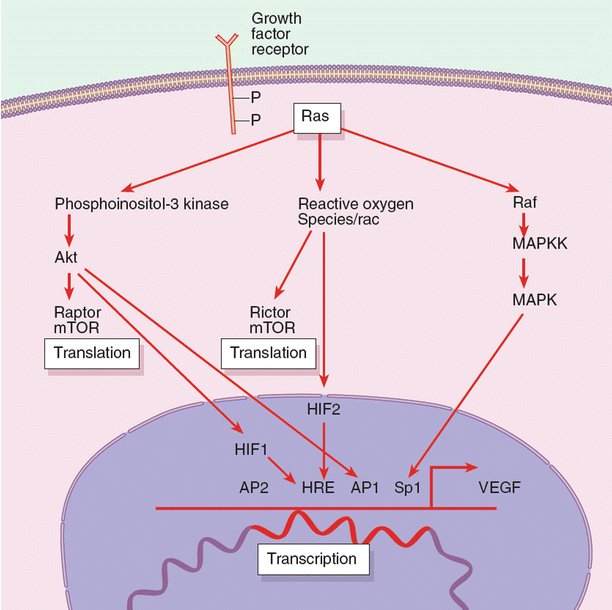

Fig. 11.2
Summary of molecular pathways that lead to vascular endothelial growth factor (VEGF) gene expression. As indicated, there are a number of signaling pathways that can activate VEGF transcriptions. HIF, hypoxia-inducible factor; MAPK, mitogen-activated protein kinase; MAPKK, mitogen-activated protein kinase kinase; mTOR, mammalian target of rapamycin; P, phosphate
The major angiogenesis stimulators in the skin are VEGF and bFGF, and the major inhibitor is interferon-α (IFN-α), the latter being a widely prescribed adjuvant therapy for melanoma. Both the major stimulants and inhibitors are produced by keratinocytes, helping to provide a barrier between vasculature and the epidermis. When there is contact between the two (barrier disruption), VEGF is increased; when there is occlusion, VEGF is decreased. This lends credence to occlusional therapies.
The Importance of Stem Cell Recruitment in Angiogenesis
The reservoir of endothelial cells for neovascular development is of two sources: local recruitment of endothelial cells, and differentiation of quiescent mesenchymal cells. Exposure of cells to cytokines and other stimuli leads to upregulation of adhesion molecules, which bind immune effector cells; such cells can migrate to sites of infection and inflammation. Withdrawal of the stimulus will lead to apoptosis of the neovasculature. Persistent endothelial stimulation, either through angiogenic growth factors, loss of endogenous factors, or mutations, will cause cells to resist physiologic apoptosis. Recall that mutations involve activation of the phosphoinositol-3-kinase (PI3K)/akt pathway; such activation accounts for neovascular formation in humans.
The recruitment of bone marrow cells is imperative for the success of vascular formation. This notion explains some observations in cancer therapy. A large group of natural products, known as chemo-preventive agents, prevent cancer development but have little effect on advanced neoplasms. Xenograft models can help select for angiogenesis inhibitors that block mesenchymal recruitment, though tumors persist in recruiting local endothelial cells. While an effective treatment may require inhibition of both pathways, recognition of the tumor’s dynamic ability to switch signaling pathways may even render recruitment-inhibiting therapies ineffective.
The ligands responsible for endothelial stem cell recruitment in ischemic tissues and tumors likely start with high expression of stromal cell-derived factor 1 (SFD-1). CXCR4 (also known as fusin is a CXC chemokine receptor 4) is the SDF1 receptor, and blockade of CXCR4 is a promising new course of therapy that has been shown to block revascularization of ischemic tissue. Matrix metalloproteinase 9 (MMP-9) mobilizes the small kit ligand (sKITL). Mobilization of sKITL could be clinically useful in the treatment of acute myocardial infarction.
Recognition by the clinician and pathologist of certain signaling markers could be vital in the early detection of aberrant cells. Angiopoietin-1 (ang1) and angiopoietin-2 (ang2) have antagonistic effects: the former binds Tie-2 to inhibit vascular permeability, while the latter binds Tie-2 to stimulate vascular permeability. Hemangiomas of infancy and the hemangioma-like verruga peruana both show elevated levels of ang2. Work by Arbiser et al. suggests that hemangiomas arise from an unknown event, causing rapid stem cell recruitment from bone marrow (Fig. 11.3). A similar situation likely exists in high-malignancy tumors. Thus early detection of alterations in ang2 expression could be developed as a diagnostic tool for early detection of tumors.
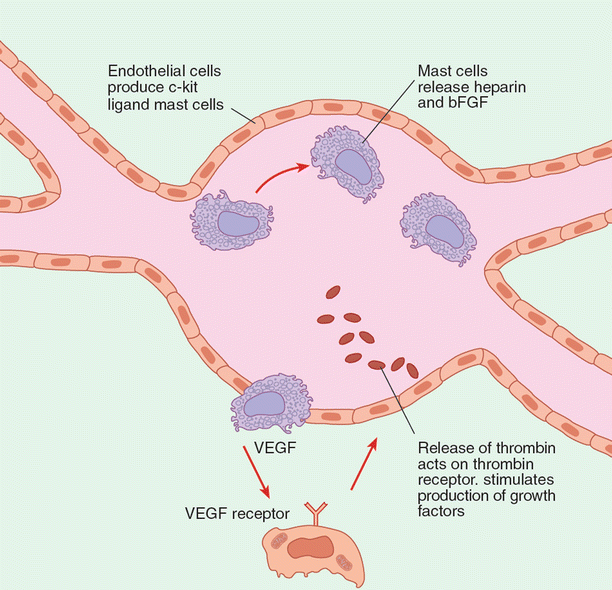

Fig. 11.3
A model for angiogenesis in hemangiomas. This model demonstrates interaction between endothelial cells. Most cells and stem cells that are recruited from bone marrow are induced to differentiate into endothelial cells
Hemangiomas exhibit a characteristic natural involution after several years. This is likely due to a decreased VEGF expression leading to apoptosis. This parallels the apoptosis observed in endothelial cells during menstruation upon lessening of estrogen signaling. This fate of the hemangioma illustrates the potent response toward removal of proangiogenic, specifically VEGF, signaling.
The beta blocker propranolol was accidently discovered to cause hemangioma regression, and has become the treatment of choice for large hemangiomas of infancy [12, 23–25]. Thus, it is possible that hemangiomas can be inhibited by beta blocker independent modes of action, and that beta blockade per se is not essential to the activity of propranolol for hemangiomas.
Inflammatory Angiogenesis
Inflammatory skin conditions can be separated into two categories. Those expressing a predominance of interferon gamma (IFN-γ), interleukin-12 (IL-12), and interferon-inducible protein 12 (IP-12) are classified as Th1. Psoriasis is considered Th17+, with IL-17 being a mainstay of inflammation. Those expressing predominantly IL-4, -5, -6, and -10 are classified as Th2, such as systemic inflammatory infiltrates of lymphocytes, mast cells, granulocytes, and macrophages. Both conditions show excess angiogenesis (despite the presence of antiangiogenic IL-12 in Th1 disorders), showing the potential utility of angiogenesis inhibitors for treatment, not merely of malignant neoplasms, but also in a wide range of dermatologic disorders.
The Role of Angiogenesis in Major Skin Disorders
Acne Vulgaris
Acne is the most common cutaneous disorder in the United States. This disorder accounts for over 10 % of all patient encounters with a primary care physician. While the number of cases of acne vulgaris in adolescents has remained relatively stable over the past decade, the number of cases of adult-onset acne is increasing. The majority of debilitating effects of acne are psychological, with embarrassment and anxiety being among the top reported symptoms. Scarring is not uncommon and contributes to the lifelong effects of a moderate to severe case.
The role of MMPs in acne has been somewhat unclear in recent work, though it appears that they are involved in acne progression. The source of MMPs in acne appears to be keratinocytes [26] or neutrophils.
Treatment of acne includes both topical and systemic therapies. For treatment of noninflammatory comedones, topical retinoids such as tretinoin, adapalene, and tazarotene are often prescribed. Salicylic acid also has proven comedolytic activity. The most prominent therapy, both over-the-counter and prescription-based, is topical benzoyl peroxide. Some patients experience increased inflammation due to the presence of Propionibacterium acnes in sebaceous follicles. Treatment of such cases usually involves topical antibiotics such as clindamycin or erythromycin. It should be noted that to lessen the opportunities for the generation of antibiotic-resistant bacteria, treatments should be coupled with benzoyl peroxide [27, 28]. In severe cases of inflammatory acne, patients can be prescribed system isotretinoin. There are prescribing restrictions for this drug that are left to the discretion of the physician. Blue light and laser therapy are expensive and unproven treatments for acne should be avoided pending further data.
Psoriasis
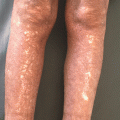
Psoriasis is a common cutaneous disorder characterized by erythematous papules and silvery-scaled plaques. Though not entirely understood, the pathophysiology of clinical psoriasis tends to result from hyperproliferation and abnormal differentiation of epidermal keratinocytes, accompanied by inflammatory cell infiltration and vascularization. This remodeling resembles a prolonged wound response, with many reparative and remodeling processes being utilized [28] (Fig. 11.4).
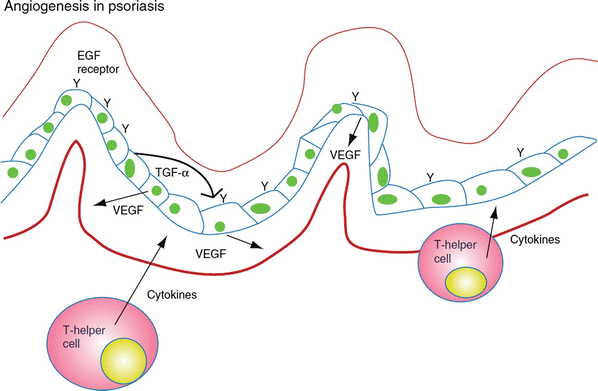

Fig. 11.4
Psoriasis is an example of chronic inflammation that drives excessive angiogenesis. It is a result of T cells and keratinocyte interactions, resulting in VEGF production, which then impacts endothelial cells
To follow the wound healing analogy presented by Nickoloff, et al. [28], tumor necrosis factor (TNF), VEGF, IL-23, and transforming growth factor (TGF) are present in high levels in psoriatic tissue. Though these cytokines are present in healthy skin, the exaggerated angiogenic response and epidermal thickening draws a definite line between healthy epidermal/dermal signaling and pathologic psoriatic skin. Genetic susceptibility plays a major role, as evidenced in the Koebner phenomenon, which describes an outbreak of psoriatic lesions on genetically susceptible patients upon mild trauma applied to the skin. Expression of TGF is a relevant early event in the formation of psoriatic lesions. Such expression leads to upregulation of VEGF, which promotes angiogenesis and changes in blood vessel morphology. In fact, elevated levels of VEGF are noted in lesional keratinocytes in psoriasis [29]. Accordingly, the angiopoietin (Ang)-Tie signaling pathway is activated in psoriasis, leading to vascular remodeling, formation, and invasion. To view the whole picture, VEGF expression leads to increased vascular permeability and capillary diameter. Once this angiogenic “switch” is turned, alterations in ang1/ang2 expression allow for increased vascular proliferation. Tumor necrosis factor provides differential direct regulation of the Tie2 signaling pathway. Tumor necrosis factor and ang1/ang2 allow for vascular survival and maintenance during this proliferative phase [30].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree