Fig. 9.1
The electrochemical transdermal device (ECTD). (a) Schematic cross section through the electrochemical patch. A external voltage source, B hydrogel, C spacer, D backing foil, E adhesive layers, F electrodes, G adhesive layers, H foam ring and I release liner (Figure reproduced with permission from John Wiley & Sons from Schröder et al. (2012a)). (b) Photograph of device taken from top surface to show external electrode contacts
The pulsatile nature of drug release from the ECTD makes it potentially suitable as an on-demand delivery system. A cursory viewing of Fig. 9.1a shows that the ECTD is not an iontophoresis-type device (Guy et al. 2001), because the skin membrane is not a part of the electric circuit existing between the two electrodes. This is a potential advantage of the ECTD over iontophoresis, since skin irritation is not expected to be a problem because voltage application is only intermittent and of short duration (≤60 s). The length of the lag phase between voltage application and incipient increase in flux is short, i.e. 2 h. The time from voltage application to maximum flux enhancement is, however, longer than is achievable with an iontophoretic device.
9.3 Previous Device from US Patent 5 533 995
It is instructive to start with a description of a transdermal device given in US Patent 5 533 995. The laminated structure of this device is illustrated in Fig. 9.2 taken from the patent description. It comprises a drug reservoir layer (Roy and Flynn 1990) composed preferably of agar or carrageenan. This drug reservoir is sandwiched between two electrodes (Sathyan et al. 2005; Schröder et al. 2012a), the lower of which is skin-facing and is permeable to the drug or functions as a gate of varying permeabilities. Between the skin-facing electrode and the skin, there is an optional transit chamber (Schröder et al. 2012b). Continuous application of an electrical potential results in a current of between 0.5 and approximately 5 mA that produces an enhancing effect on the flux of the model drug physostigmine through the excised human skin. The authors of the patent recognized that this enhancement is not caused by iontophoresis, as the skin is not part of any electrical circuit. They suggested that an ‘active’ transport process of the drug within the gel takes place through the skin-facing electrode to the skin surface. The mechanism of this active transport was considered to be electrophoresis: the electric current produces movement of ionized physostigmine through the gel resulting in a build-up of drug concentration in the vicinity of the porous skin-facing electrode. The enhancing effects achieved required, however, a continuous application of voltage over times between 3 and 6 h.
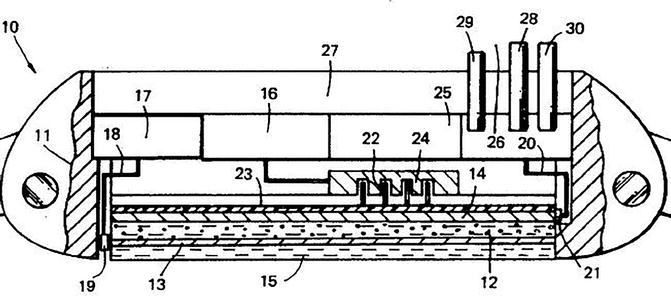

Fig. 9.2
Structure of passive transdermal device from US Patent 5 533 995. The following parts are relevant for a comparison with our ECTD. 12 drug reservoir, 13 + 14 electrodes, 15 transit chamber
9.4 Electrochemical Mechanism of the ECTD
The ECTD differs substantially from that described in US Patent 5 533 995. Its manufacture on the laboratory scale has been fully described before (Schröder et al. 2012a). The hydrogel is formed using hydroxypropyl cellulose (HPC) (see Table 9.1) that contains either 1 % w/w fentanyl base (# 1) or 1.57 % w/w fentanyl citrate (# 2). The hydrogel’s pH is adjusted by adding suitable quantities of HCl and NaOH. The in vitro release/permeation behaviour of fentanyl from the ECTD was examined using excised mouse skin membranes fitted in a tailor-made diffusion cell (Schröder et al. 2012a). In the first experiment shown in Fig. 9.3a, the hydrogel # 1 was used at pH 4.0 together with two Ag-coated stainless steel electrodes. Different patches were examined whose attributes are given in the legend to the applicable Figure. Patch A was used in ‘passive’ mode with no voltage being applied, and there was consequently a slow release from the patch and a low permeation through the skin membrane. To patch C, a 2 V pulse of 60 s duration was applied at t = 18.5 h and also t = 42.5 h. These times were selected to enable clear recognition of the effects of voltage application. This produces a sharp and lasting increase in the permeated drug mass m(t) – an enhanced skin permeation that is maintained over many hours, despite the short duration of the voltage application. With patch B, an impulse of 1 V for 60 s at t = 18.5 h produces no permeation enhancement. Enhancement requires 2 V when replied at t = 42.5 h. There is therefore a minimum voltage somewhere between 1 and 2 V that is required to cause enhanced permeation.
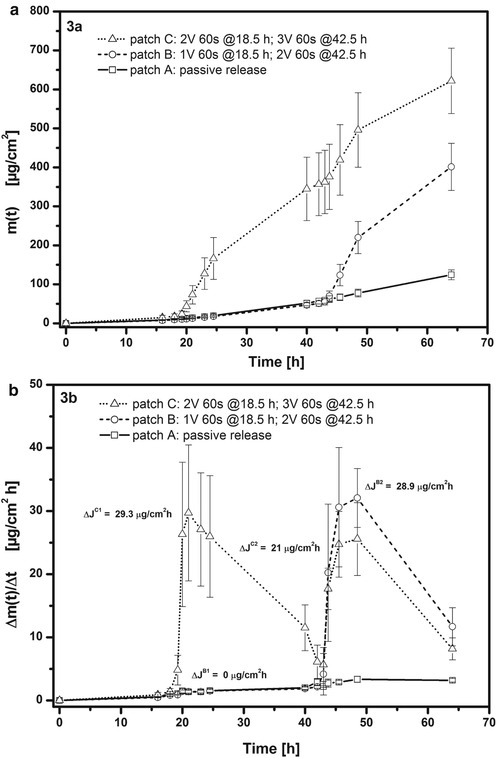
Table 9.1
The two hydrogel formulations
Substance | #1 [% w/w] | #2 [% w/w] |
|---|---|---|
Fentanyl base | 1.0 | – |
Fentanyl citrate | – | 1.57 |
HPC | 2.5 | 2.5 |
HCl (5 % w/w) | 0.75 | 0.75 |
NaCl | 0.1 | 0.1 |
Sorbic acid | – | 0.1 |
Water | ad 100 | ad 100 |
NaOH | q.s. for pH adjustment |

Fig. 9.3
Measured release/permeation profiles of fentanyl from ETCD (hydrogel # 1) through excised mouse skin. Patch A passive release (no voltage application); patch B 1 V for 60 s at t = 18.5 h and 2 V for 60 s at t = 42.5 h; patch C 2 V for 60 s at t = 18.5 h and 3 V for 60 s at t = 42.5 h. (a) Plots of cumulative amount of fentanyl in acceptor, m(t), versus time. (b) Plots of fentanyl flux, Δm(t)/Δt, into acceptor versus time (Reproduced with permission from John Wiley & Sons from Schröder et al. (2012a))
Figure 9.3b shows the first derivative of each of the plots in Fig. 9.3a which is the flux of the drug into the acceptor, Δm(t)/Δt in μg cm−2 h−1. The 1 V application to patch B produces no change in flux, whereas the 2 V for 60 s applied later at t = 42.5 h increases flux by some 29 μg cm−2 h−1. This is the same value as seen with patch C after a first 2 V application for 60 s at t = 18.5 h. The second voltage application to patch C at t = 42.5 h is of 3 V for 60 s, yet the flux enhancement is only some 21 μg cm−2 h−1 despite the higher voltage. Neither this weakening of flux enhancement on repeated voltage application nor the minimum voltage of 1–2 V required to initiate flux enhancement indicates an iontophoresis mechanism. Further evidence against iontophoresis as an enhancement mechanism is given in Fig. 9.4 which shows the effect of substituting fentanyl base (hydrogel # 1) with fentanyl citrate (hydrogel # 2). The iontophoretic mobility of the latter should be higher than that of the fentanyl base, yet patch D (hydrogel # 2) shows a substantially lower flux enhancement than seen with the first voltage application to patch C in Fig. 9.3b. In addition, the increase to 9 V (patch E) gives on first application at t = 15.5 h a stronger flux enhancement than with 2 V (patch D), but on second application, no further flux enhancement effect was observed.
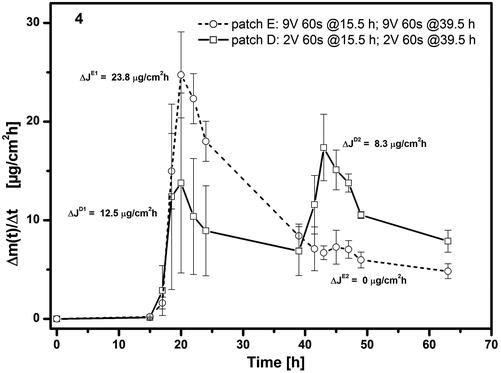

Fig. 9.4
Effect of substituting fentanyl citrate (hydrogel # 1) with fentanyl base (hydrogel # 2) on release/permeation of fentanyl from ECTD through excised mouse skin. Patch D (hydrogel # 2): 2 V for 60 s at both t = 15.5 h and 39.5 h. Patch E (hydrogel # 1): 9 V for 60 s at both t = 15.5 and 39.5 h. Plots of fentanyl flux, Δm(t)/Δt, into acceptor versus time (Reproduced with permission from John Wiley & Sons from (Schröder et al. 2012a))
The mechanism of working of the ECTD appears to be a straightforward electrolysis at the two silver electrodes of the water held in the hydrogel. This will occur once the potential difference across the electrodes is higher than approximately 1.5 V at 25 °C (Weast R. Handbook of Chemistry and Physics et al. 1985). The electrolysis reaction generates hydroxyl ions at the cathode and hydronium ions at the anode which in the case of Ag electrodes will alter the pH of the hydrogel. Studies on other hydrogels (Murdan 2003) have shown that the generation of hydroxyl ions at the cathode evidently causes deprotonation of a weak base dissolved in the hydrogel in the vicinity of the cathode. Measurement of the bulk hydrogel’s pH in the ECTD indeed showed that this increased from 4 to >7 during the course of the release/permeation experiments with patches B–E, but not with the ‘passive’ patch A. As this pH shift affects the whole of the hydrogel layer, we expect the dissolved fentanyl to become less ionized and have therefore increased permeability through the adjacent skin membrane (Roy and Flynn 1990). The advantage of this electrochemical mechanism over iontophoresis is the use of a brief (60 s) voltage pulse rather than constant current application over hours. Additionally the lag phase (τ) between voltage application and increase in flux is short, i.e. 2 h. The duration between the time point of voltage application and that of maximum flux into the receptor is 2.5 and 6 h (see Figs. 9.3b and 9.4) as shown with mouse skin in vitro.
9.5 Development of a Suitable Hydrogel Formulation for the ECTD
Hydroxypropyl cellulose was selected as a gelling agent (see Table 9.1) because it is non-electroresponsive, i.e. it is non-ionizable and as such should show no voltage-induced changes in its properties such as swelling or de-swelling (Murdan 2003; Scranton et al. 1995). Increase in the HPC concentration in hydrogel # 2 from 1.5 % w/w to 3.5 % w/w has no effect on the flux enhancement behaviour on application of 2 V for 60 s after t = 1 h followed by 3 V for 45 s after t = 23.5 h (Fig. 9.5). Both the maximum flux and lag time are invariant, despite the different viscosities of the gels of 0.9–5 Pa∙s (shear rate = 107 s−1). The electrochemical reaction at the electrodes that is necessary to induce flux enhancement is not therefore dependent on the gelling agent concentration in the range examined. HPC used at the concentration of 2.5 % w/w is therefore a suitable gelling agent to produce adequate gelation without hindering the magnitude of flux enhancement.
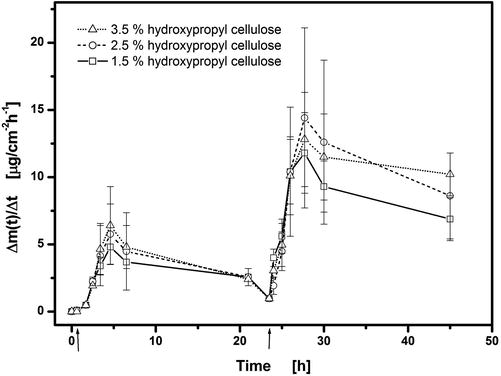

Fig. 9.5
Effect of concentration of hydroxypropyl cellulose (HPC) on release/permeation of fentanyl (hydrogel # 2) from ECTD through excised mouse skin. Voltages of 2 V for 60 s at t = 1 h and 3 V for 45 s at t = 23.5 h were applied. Plots of fentanyl flux, Δm(t)/Δt, into acceptor versus time
The presence of 0.1 % w/w sorbic acid as a preservative in the hydrogel (see Table 9.1) was necessary to meet the antimicrobial efficacy criteria A and B of the European Pharmacopoeia. Sorbic acid has bacteriostatic activity in a concentration of 0.1 % w/w at pH values below 4.5 (the initial hydrogel pH is 4.0). Its presence has no influence on flux enhancement after application of 2 V for 45 s at t = 22.75 h followed by 3 V for 60 s at t = 46.5 h (Fig. 9.6). Neither the maximum flux nor lag time shows any dependence on sorbic acid content.
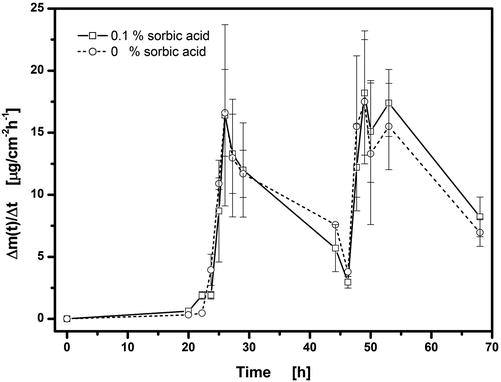

Fig. 9.6
Influence of added sorbic acid to hydrogel on release/permeation of fentanyl (hydrogel # 1) through excised mouse skin. Voltages of 2 V for 45 s at t = 22.75 h and 3 V for 60 s at t = 46.5 h were applied. Plots of fentanyl flux, Δm(t)/Δt, into acceptor versus time
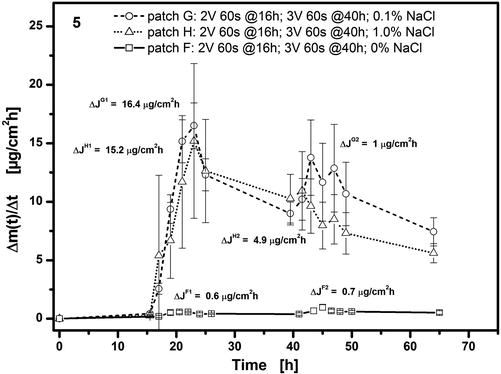
The presence of a source of chloride ions in the hydrogel is essential to achieve any flux enhancement on voltage application. This is illustrated in Fig. 9.7 where in the absence of sodium chloride (patch F) no flux enhancement effect is observed after voltage applications of either 2 V or 3 V. At the end of the experiment, this patch was disassembled, and the pH measured in the gel had remained unchanged at a value of pH 4.0. Without chloride ions, the electrolysis reaction will generate and release H3O+ at the anode which counters any pH increase caused by release of OH− at the cathode. An increase in pH is, however, necessary to produce any flux enhancement; so the lack of chloride ions fully inhibits the work of the ECTD. In the presence of chloride ions (patches G and H), the silver anode is oxidized to insoluble silver chloride during electrolysis which is deposited on the anode surface. The resulting discoloration of the anode can be directly observed in the ECTD shown in Fig. 9.8a taken after two voltage applications each of 2.5 V for 60 s at t = 16 h and t = 40 h. There is therefore no release of H3O+ from the anode into the hydrogel and hence no hindrance of the pH increase generated by the cathode and necessary for flux enhancement. Figure 9.7 also illustrates that a level of 0.1 % w/w NaCl in the hydrogel is evidently sufficient to ensure this mechanism and hence produce flux enhancement.