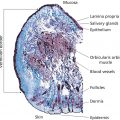
M. Amanda Jacobs1, Randall Roenigk2, and Cassondra A. Ellison1 1 Geisinger Health Systems, Danville, PA, USA 2 Department of Dermatology, Mayo Clinic, Rochester, MN, USA Superficial chemical peels involve the application of a chemical peeling agent to the skin, resulting in destruction of the epidermis. The peel may have effects anywhere from the stratum corneum to the basal cell layer. There are many factors that affect the depth of the peel including the peeling agent and the technique of application. Chemical peels create a controlled wound which the body heals at a later point of time. Permanent histologic changes can be seen after a series of superficial peels. Superficial chemical peels are generally safe and can be used on all Fitzpatrick skin types. The peeling agent only affects the epidermis and so this procedure is only indicated for superficial processes such as mild photoaging, superficial dyschromias (melasma, lentigines, and post‐inflammatory hyperpigmentation [PIH]), acne, and actinic keratoses [1]. Typically, a series of peels is required for the best clinical response. Multiple factors influence the depth of a given peel, including chemical agent selection and technique of application (Table 49.1). Each of these factors should be considered when selecting a superficial peel for a patient. Superficial chemical peels injure and rejuvenate the epidermis; however, histologic changes can also be seen in the dermis, including collagen formation in the papillary dermis. Repeated application of α‐hydroxy acids (AHA) to the skin surface resulted in a 25% increase in epidermal thickness as well as increased acid mucopolysaccharides and increased density of collagen in the papillary dermis [2]. In a superficial peel, chemical agents are applied to the skin to create a wound by destroying the epidermis. Recovery time is minimal (24–48 hours). There are multiple chemicals that fall into this category (Table 49.2). These acids have been used on the skin for centuries. Ancient Egyptian women would bathe in sour milk (containing lactic acid) to smooth the skin [3, 4]. They are naturally occurring in sugarcane, sour milk, and various fruits. When applied to the skin, they decrease corneocyte adhesion above the granular layer, reduce the number of desmosomes, and result in desquamation [2, 4]. The application is typically painless, although some mild stinging may occur. These acids require neutralization with sodium bicarbonate or dilution with water. Prolonged contact with the skin may result in uneven penetration and deeper peel depths being achieved [1]. Table 49.1 Variables that affect the depth of a superficial peel. Table 49.2 Characteristics of various superficial chemical peeling agents. a White precipitate can occur as vehicle evaporates, does not represent true frost. b 4 g resorcinol, 14 g salicylic acid, 14 g lactic acid in alcohol solution. This is a β‐hydroxyl acid. It is a lipophilic acid that allows desquamation of the upper layers of the stratum corneum without inflammation [1, 5]. This peeling agent can be safely used on all skin types, even those prone to PIH. This agent also has keratolytic and comedolytic qualities making it ideal for acne patients. It also enhances the penetration of other acids and is often used in combination peels (such as Jessner’s solution). A mild–moderate burning sensation occurs on application. Typically, this is easily managed with a cool fan. The acid causes superficial anesthesia and so this quickly dissipates after a minute or more. The acid should be left on the skin for about 3–5 minutes. This is a self‐neutralizing acid but is typically washed off the skin after 5 minutes with plain water. A white precipitate will be seen on the skin after about 1 minute from evaporation of the vehicle (Figure 49.1). This is not a true frost and does not indicate depth of penetration. Desquamation will begin 2–3 days after the peel and continue for 7 days [1, 4, 5]. Salicylism is possible with this peel, although unlikely. Care should be taken to avoid peeling large areas at the same time. Patients may experience tinnitus, dizziness, and headache. The symptoms are transient and self‐resolving [6]. Increased water intake may improve symptoms more quickly. Figure 49.1 White discoloration of the skin seen during a salicylic acid peel caused by evaporation of the vehicle and precipitation of salicylate on the skin. The penetration of this solution is limited to the epidermis. The peel results in separation of the stratum corneum and upper epidermal edema [1]. Application is best with a sable hair brush. Wrung‐out gauze pads may also be used. Typically, two to three coats are applied. The skin will first become erythematous and then a white, powdery appearance will emerge. This is caused by precipitation of the chemical compounds onto the skin with vehicle evaporation. This is not equivalent to a “frost” which indicates depth of peeling in a trichloroacetic acid (TCA) peel. Complications are rare with this type of peel because of the limited penetration of the peeling solution. This product has keratolytic, antimicrobial, and sebostatic properties. It is a small molecule with a low pK a
CHAPTER 49
Superficial Chemical Peels
Definition
Physiology
Indications
Depth of peel
Histologic changes
Formulation
α‐Hydroxy acids (glycolic, lactic, malic, oxalic, tartaric, and citric acid)
Peeling agent and concentration
Technique of application (number of coats and pressure of application)
Duration of contact with the skin (prior to neutralization)
Anatomic location
Pretreatment regimen
Peeling agent
Indication
Activity
Neutralization
Frosting
Unique properties
Side effects
α‐Hydroxy acid (e.g., glycolic acid)
Mild skin damage; dyschromias
Low concentrations (5–10%) produce corneolysis; high concentrations (50–70%) induce epidermolysis
+
−
Deeper peel may be achieved with prolonged skin contact
β‐Hydroxy acids (e.g., salicylic acid)
Mild skin damage; dyschromias; acne
Keratolytic, comedolytic, desquamation of upper SC enhances other peeling agents
−
−a
Burning with initial application, then acid becomes an anesthetic
Transient salicylism may occur
Jessner solutionb
Mild skin damage; dyschromias
Corneolysis (increasing effect of other keratolytic agents), dermal edema
−
−a
Intense burning with application 2–3 coats applied
Pyruvic acid
Mild skin damage; dyschromias; acne
Keratolytic, sebostatic, antimicrobial
+
−
Intense burning with application
Newer formulation used for superficial peels
Trichloroacetic Acid (TCA, 10–25%)
Fine wrinkles and acne scars; dyschromias
Coagulation of epidermal proteins
−
+
True frost forms, intensity correlates with depth of injury
Burning with application Erythema for several days
Tretinoin
Fine wrinkles, acne
Not destructive; increases cell turnover
−
−
Painless
Produces strong erythema Fine white flakes with peeling
Discolors skin (yellow)
Must remain on skin for 6 hours
Decomposed by UV light
Resorcinol
Fine wrinkles
Weakens hydrogen bonds of keratin
−
−
Daily application for 3 days; time consuming
Irritant or contact allergy possible
Continued use associated with thyroid dysfunction
Solid carbon dioxide
Acne
Freezes and destroys epidermis
Block of dry ice dipped in acetone/ethanol solution
Cold sensitivity
Used to treat acne
Salicylic acid (ortho‐hydroxybenzoic acid)
Jessner solution
Jessner solution (resorcinol 14%, lactic acid 14%, and salicylic acid 14% in alcohol)
Pyruvic acid (α‐keto acid)
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree