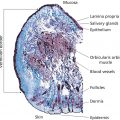
Barbara A. Green1, Eugene J. Van Scott2, and Ruey J. Yu3 1 NeoStrata Company, Johnson and Johnson Consumer Inc., Skillman, NJ, USA 2 Private Practice, Abington, PA, USA 3 Private Practice, Chalfont, PA, USA Research on aging today is motivated by the belief that innate degenerative processes of aging, and aging itself, can be modulated, prevented, and perhaps reversed. Even though a complete understanding of the mechanisms of aging may not be known, improvement in clinical appearance and function of the skin with anti‐aging measures is a signal that modulation of aging and/or degenerative processes has probably occurred. After many years of research and clinical use, hydroxyacids (HAs) have been shown to have biologic importance and clinical value for both younger skin and older skin with a variety of hyperkeratotic and aging‐related conditions. This chapter covers HAs, including AHAs, BHAs, PHAs, aldobionic acids (bionic acids), and AMAs. These compounds positively impact skin morphology and function, naturally playing physiologic roles in promoting normalcy and defending the skin against endogenous and exogenous adversities. They provide measurable clinical and anti‐aging benefits that can be considered both preventative and corrective over the course of time. HAs may be divided into five groups based on their chemical structures: AHAs, BHAs, PHAs, bionic acids, and AMAs. The AHAs are the most widely studied and commercialized ingredients within the HA family. They are the simplest of the HAs, consisting of organic carboxylic acids with one hydroxyl group attached to the alpha position of the carboxyl group. Both the hydroxyl and carboxyl groups are directly attached to an aliphatic or alicyclic carbon atom. As a result, the hydroxyl group in the AHA is neutral and only the carboxyl group provides acidity to the molecule, a property that distinguishes the AHAs from aromatic HAs such as salicylic acid () as described below. Many AHAs are present in foods and fruits, and therefore are called fruit acids (Table 42.1). Glycolic acid, the smallest AHA, occurs in sugar cane and citric acid is found in lemon juice at a concentration of 5–8%. Aralkyl alpha‐hydroxyacids (ARAHAs) contain a phenyl group as a side chain substituent. In contrast to SA, the hydroxyl group in ARAHAs are attached to the alkyl carbon atom instead of aromatic phenyl carbon atom. The phenyl group changes the solubility profile of the ARAHA providing increased lipophilicity over conventional water‐soluble AHAs and can be used to target oily and acne‐prone skin. Examples include mandelic acid (phenyl glycolic acid), benzilic acid (diphenyl glycolic acid), and 3‐phenyl lactic acid. Solubility of these more lipophilic compounds can vary, for instance benzylic acid is not soluble in water, but only in a lipid solvent such as ethanol and acetone, whereas mandelic acid is soluble in both water and ethanol. Table 42.1 Examples of hydroxyacids. The BHAs are organic carboxylic acids with one hydroxyl group attached to the beta position of the carboxyl group. Both the hydroxyl and carboxyl groups are attached to two different carbon atoms of an aliphatic or alicyclic chain rendering the hydroxyl group neutral in nature. Some BHAs are present in body tissues as metabolic intermediates and energy source. For example, β‐hydroxybutanoic acid is produced by the liver and utilized by skeletal and cardiac muscle as an energy source. In contrast to the water‐soluble β‐hydroxybutanoic acid, tropic acid is derived from a plant source, and is a lipid‐soluble BHA. For the most part, BHAs have yet to be commercialized in skincare mainly because of a lack of commercial supply and high cost. Some AHAs are also BHAs when the molecule contains two or more carboxyl groups. In this case, the hydroxyl group is in the alpha position to one carboxyl group, and at the same time is in the beta position to the other carboxyl group. For example, malic acid (apple acid) with one hydroxyl group and two carboxyl groups is both an AHA and a BHA. In the same manner, citric acid contains one hydroxyl group relative to three carboxyl groups and is both an AHA to one carboxyl group and a BHA to the other two carboxyl groups. These ingredients have been commercialized in skincare formulations to adjust pH and to deliver antioxidant and anti‐aging benefits. The PHAs are organic carboxylic acids that possess two or more hydroxyl groups in the molecule. The hydroxyl and carboxyl groups are attached to the carbon atoms of an aliphatic or alicyclic chain. All the hydroxyl groups in the PHA are neutral, and only the carboxyl group accounts for its acidity. Although hydroxyl groups may be attached to several positions of the carbon chain, in order to be both an AHA and PHA, it is essential that at least one hydroxyl group be attached to the alpha position. Many PHAs are endogenous metabolites or intermediate products from carbohydrate metabolism in body tissues. Both galactonic acid and galactonolactone are derived from galactose, which is an important component of glycosaminoglycans. Gluconic acid and gluconolactone are important metabolites formed in the pentose phosphate pathway from glucose during the biosynthesis of ribose for ribonucleic acid. Gluconolactone is the most widely studied and commercialized skincare ingredient among the PHAs (Table 42.2). The aldobionic acids, also called bionic acids, consist of one carbohydrate monomer chemically linked to a PHA via a stable ether linkage; examples are lactobionic acid (galactose/gluconic acid) and maltobionic acid (glucose/gluconic acid) (Figure 42.1). The bionic acid is commonly obtained from its disaccharide through chemical or enzymatic oxidation. For example, lactobionic acid is obtained from lactose and maltobionic acid from maltose. In general, the bionic acids have a larger molecular size and weight than most PHAs because of the additional sugar unit; however, at 358 Da (lactobionic acid and maltobionic acid), these molecules remain small enough to penetrate skin. AMAs such as SA and gallic acid are derived from benzoic acid, which has been used in combination with SA as Whitfield’s ointment (e.g. 6% benzoic acid/3% SA) for hyperkeratotic conditions and fungal infections. SA is 2‐hydroxybenzoic acid and may be called an HA within a broad definition; however, its effects on skin differ from those of the AHAs, BHAs, PHAs, and bionic acids. SA is a conventional keratolytic agent which desquamates corneocytes, layer by layer from the top downward [1]. In contrast, AHAs and BHAs appear to act at the innermost layers of the stratum corneum, the stratum compactum, near the junction with stratum granulosum [2, 3]. Moreover, AHAs, PHAs, and bionic acids have been shown to stimulate biosynthesis of dermal components and increase dermal skin thickness upon topical application, whereas SA has been shown to decrease dermal skin thickness [3–7]. SA is an approved topical drug for the treatment of acne in accordance with the United States Food and Drug Administration over‐the‐counter (OTC) acne drug products monograph. Its use in cosmetics may be less desirable relative to other HAs due to dermal thinning effects. Table 42.2 Antiaging, antioxidant and skin protection benefits of PHAs. Figure 42.1 Maltobionic acid, a bionic acid. As individual compounds, HAs differ broadly in physicochemical properties. Some HAs are very small molecules such as glycolic acid and lactic acid, and some are larger molecules such as lactobionic acid. Most HAs are white crystalline at room temperature such as glycolic acid, malic acid, tartaric acid, citric acid, mandelic acid, and benzilic acid, but a few are liquid such as lactic acid. Most HAs are soluble in water, but some are also soluble in lipid solvents. Some HAs are soluble only in lipid solvents. Certain physicochemical properties of HAs are discussed herein. Mandelic acid is an ARAHA with increased lipophilicity over conventional water‐soluble AHAs, making it particularly well suited to be used in a sebum‐rich environment associated with oily or acne prone skin. Sebum lipids, such as squalene and wax esters, were evaluated by measuring the neutral lipid production in human sebocyte cultures and showed mandelic acid (0.05%) significantly reduced neutral lipid production (p < 0.05 versus untreated) with a 41% inhibition in production compared to the positive control. The water‐soluble glycolic acid (0.05%) did not demonstrate these same benefits [8]. Mandelic acid would be an AHA of choice with these added benefits in targeting oily and acne‐prone skin. In contrast to AHAs and BHAs, PHAs and bionic acids are strongly hygroscopic and can attract and bind water similarly to other polyol compounds such as glycerin. The bionic acids are so strongly hygroscopic that they form a gel matrix when their aqueous solution is evaporated at room temperature. The transparent gel thus obtained retains certain amounts of water, forming a clear gel matrix. The amount of water retention depends on the individual bionic acid. For example, maltobionic acid can form a clear gel film containing 29% water complexed with maltobionic acid molecules. The formation of a gel matrix provides moisturization and may add protective and soothing properties for inflamed skin. PHAs and bionic acids are gentle and non‐irritating, and can be used to provide anti‐aging benefits to sensitive skin including patients with rosacea and atopic dermatitis, and following cosmetic procedures that weaken skin barrier function and increase skin sensitivity [9–12]. Most PHAs, bionic acids, and some AHAs and BHAs with one hydroxyl group and two or more vicinal carboxyl groups have been found to be antioxidants. The antioxidant property is readily determined by using any one of the following test methods: prevention or retardation from air oxidation of: (a) anthralin, (b) hydroquinone, or (c) banana peel. A freshly prepared anthralin solution or cream is bright yellow, and an air‐oxidized one is brownish or black. A hydroquinone solution or cream is colorless or white color, and an air‐oxidized one is brownish or black. A freshly peeled banana peel is light yellow in color, and an oxidized one ranges in color from tan, dark tan, brown to brownish black. Known antioxidants such as vitamin C and N‐acetylcysteine may be used as the positive control in these screen tests. Based on these tests, all the PHAs and bionic acids tested are antioxidants, which include ribonolactone, gluconolactone, galactonolactone, lactobionic acid, and maltobionic acid [12]. Among AHAs and BHAs, malic acid, tartaric acid, citric acid, and isocitric acid have been shown to be antioxidants. Another method to determine antioxidant, free radical scavenging properties utilizes an in vitro model of cutaneous photoaging. Compounds are assessed to determine their ability to prevent UV‐induced activation of the elastin promoter gene in skin via free radical scavenging activity. An increase in the expression of this gene causes the abnormal production of poorly structured elastin in skin, resulting in the condition known as solar elastosis – a hallmark of photoaging. In this model, free radical scavengers can reduce elastin promoter gene activation by approximately 50%; the balance of gene activation is reportedly caused by direct UV damage to cells and cellular DNA, and can only be prevented with agents that protect the skin against UV penetration such as sunscreens. Results of the study indicate that the PHA, gluconolactone, provides up to 50% reduction of gene activation, the maximum effect afforded by antioxidant/free radical scavengers. Gluconolactone is not a significant UV absorbing compound (i.e. sunscreen) and the results were therefore attributed to the compound’s ability to chelate oxidation‐promoting metals and possibly via direct free radical scavenging effects [13]. It is interesting to note that lactobionic acid is an important antioxidant chelator used in organ transplantation preservation solutions to suppress tissue damage caused by hydroxyl radicals during organ storage and blood reperfusion. In this regard, lactobionic acid reportedly inhibits hydroxyl radical production by forming a complex with Fe(II) [14]. Furthermore, both lactobionic acid and maltobionic acid have been shown to function as hydroxyl radical scavengers in the in vitro lipid peroxidation antioxidant model [15–17]. Inhibition of lipid peroxidation is vital for maintaining cell membranes and mitochondria, protecting cells against sun damage and oxidative stress, and is a measure of a substance’s antioxidant capacity.
CHAPTER 42
Clinical Uses of Hydroxyacids
Introduction
Chemical categorization and natural occurrence of hydroxyacids
Alpha‐hydroxyacids
Category
Example
Occurrence/source
Antioxidant
Alpha‐hydroxyacid (AHA)
Glycolic
Sugar cane
No
Lactic
Sour milk, tomato
No
Methyllactic
Mango
No
Citric
Lemon, orange
Yes
Aralkyl AHA (ARAHA)
Mandelic
Almonds
No
Malic
Apple
Yes
Tartaric
Grape
Yes
Beta‐hydroxyacid (BHA)
Beta‐hydroxybutanoic
Urine
No
Tropic
Plant
No
Polyhydroxy acid (PHA)
Gluconic
Skin, commercially derived from corn
Yes
Gluconolactone
Skin, commercially derived from corn
Yes
Aldobionic acid (bionic acid)
Lactobionic
Lactose from milk
Yes
Maltobionic
Maltose from starch
Yes
Aromatic hydroxyacid (AMA)
Salicylic
Ester form in wintergreen leaves
No
Beta‐hydroxyacids
Polyhydroxy acids
Aldobionic acids or bionic acids
Aromatic hydroxyacids
Polyhydroxy acids
Benefit
Gluconolactone
Lactobionic acid
Maltobionic acid
Exfoliation and cell turnover
√
√
√
Softer, smoother skin; clear pores
Pigment evening
√
√
√
Even skin tone, increased clarity, reduced spots
Increases GAGs/collagen
ProCollagen
GAGs
GAGs
Plumps and volumizes skin; GAGs hold skin’s water; Collagen increases firmness
Plumps/firms
√
√
√
Skin looks fuller and feels more elastic
Chelation
√
√
√
Traps oxidation promoting metals
Lipid peroxidation
√
√
√
Prevents oxidative damage to lipids (e.g. cell membranes, mitochondria)
Elastase inhibitor
√
Not tested
Not tested
Preserves healthy elastic tissue
MMP inhibitor
√
√
√
Preserves dermal matrix (collagen, GAGs)
Antiglycation
√
√
√
Preserves skin’s natural collagen/elastin by reducing Advanced Glycation End‐products
Barrier building
√
√
Not tested
Strengthens barrier function against irritants
Gentle, non‐irritating
√
√
√
With no sensory responses such as stinging or burning
No sun sensitivity
√
Not tested
Not tested
No increase in sunburn cells after UV irradiation
Water‐binding
√
√
√
Increases humectancy 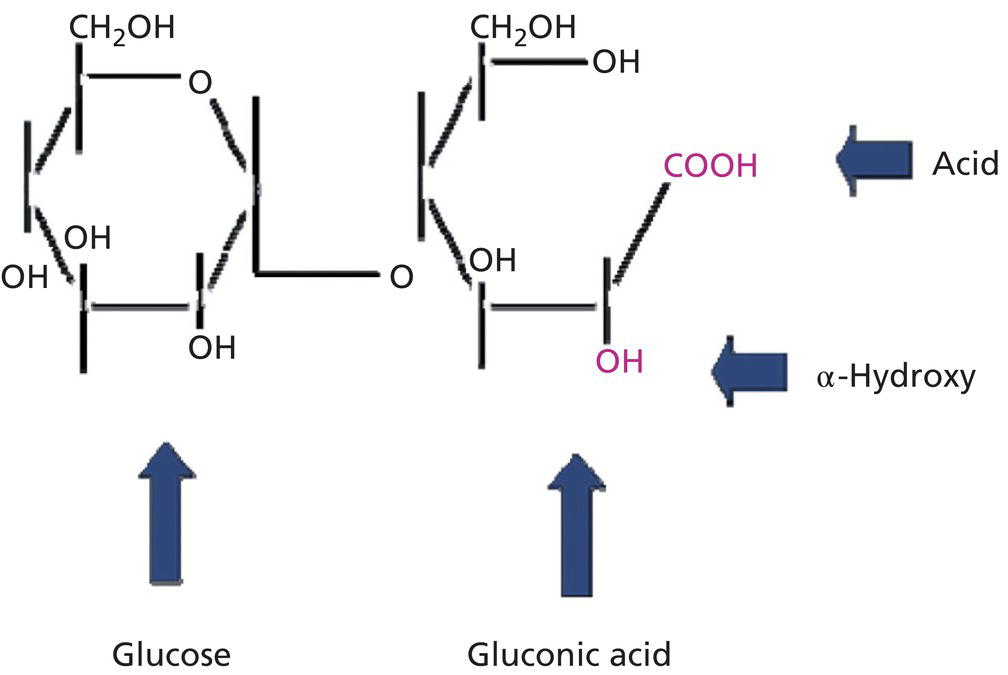
Physicochemical and biological properties distinguishing HAs
Inhibition of lipid production with lipophilic AHA
Water‐binding properties/gel matrix formation
Antioxidant properties
Antiglycation effects of PHA and bionic acids
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree