32 Embryology and Classification of Hand Differences
Summary
Congenital hand differences arise from genetic or environmental influences on the fetus during development. Embryogenesis of the upper limb occurs between the fourth and eighth weeks of gestation. Normal limb development requires a time-dependent regulated sequence of transcription factors and growth factors. It is best considered by simplification into the three axes of development: proximodistal, anteroposterior, and dorsoventral. Dysmorphology can result from disruption at any stage during upper limb development and involve any structures. The International Federation of Societies for Surgery of the Hand morphologically based classification of congenital hand differences predated current knowledge of etiology and embryological development. The Oberg–Manske–Tonkin classification attempts to incorporate this to allow clinicians and developmental biologists to communicate in a common language. It offers a framework for conveying the etiology of the anomaly while remaining flexible to new developments.
32.1 Introduction
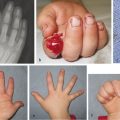
The incidence of upper limb anomalies is approximately 1 in 450 live births based on United Kingdom data. If one anomaly is identified, there is also an increased likelihood of a further non–upper limb anomaly. There is huge variety in the severity of the appearance and the functional deficit associated with a congenital hand difference with many upper limb malformations resulting in relatively little disability due to adaptive strategies. The range of congenital anomalies is vast, ranging from a barely discernible mild thumb hypoplasia to complete agenesis.
Management of the patient requires accurate assessment and diagnosis, clear communication with the family and child, and may require therapy or surgical intervention. To Properly assess such patients, a sound understanding of the pathogenesis of the anomaly, and thus of normal upper limb embryology, is required. In this chapter, we will discuss normal embryology and development of the upper limb. We will then relate this to the stages during which anomalies can develop, and finally the accepted framework for classification of such congenital anomalies.
32.2 Early Embryology
The zygote undergoes cleavage during the first week of development to produce a blastocyst, with an inner embryoblast and an outer trophoblast. In week 2, the dorsoventral (DV) axis of the embryo is established, with splitting of the embryoblast into the primary ectoderm and primary endoderm. In the third week, the primitive streak arises at the caudal end of the bilaminar germ disc, establishing the longitudinal axis of the embryo. The primitive node forms at the cranial end of the primitive streak and connects to the notochord. The notochord process is a mesodermal midline structure that induces the development of the neural plate in the ectoderm.
Gastrulation begins in the third week. During this process, the definitive endoderm, ectoderm, and mesoderm are formed, converting the embryo into a trilaminar germ disc. The primitive streak regresses, during which mesodermal cells on either side of the notochord form the somatic mesoderm. These later differentiate into the axial skeleton, voluntary musculature, and part of the skin dermis. A less pronounced lateral plate mesoderm forms and splits into a ventral layer associated with the endoderm, which forms the mesothelial covering of the viscera and heart, and a dorsal layer associated with the ectoderm, which forms the body wall lining and dermis.
32.3 Limb Bud Development
Limb bud development occurs between the fourth and eighth weeks of embryonal development, with a later prolonged growth phase after formation. The notochord expresses sonic hedgehog (SHH) between days 21 and 22, which regulates the initiation of the limb bud. The limb bud is an outgrowth of somatic mesoderm and lateral plate mesoderm into the overlying ectoderm, which forms an outer ectodermal “cap.” The upper limb buds are first noted on the lateral body wall 24 days after fertilization in the lower cervical region, at the level of C5–C8. At 26 days, a ridge of bulging ectoderm develops on the ventromedial border of the limb bud, termed the apical ectoderm ridge (AER). Underlying this is a layer of undifferentiated mesenchymal cells termed the progress zone (PZ). Both are important in ensuring proximodistal (PD) outgrowth of the limb, via messenger proteins.
The human upper limb bud can be divided developmentally into four main zones: the stylopod, the zeugopod, the mesopod, and the autopod, which correspond approximately to the arm, forearm, wrist, and hand, respectively. Positional information regarding cell destination along the PD axis is conferred based on the length of time spent within the PZ. Cells exiting the PZ early are destined to form more proximal structures such as the humerus, while those with a longer residence develop into the more distal structures of the limb. Furthermore, each set of cells exiting the PZ differentiate and express different growth factors: the future stylopod expresses MEIS-1, the zeugopod cells express OXA11 and HOXD9, mesopod cells express HOXA1, and the autopod cells express HOXA13 and HOXD10–13.
32.3.1 Axes of Development
Limb growth and differentiation progress along three axes: PD, anteroposterior (AP), and DV. The PD axis refers to elongation of the limb, the AP axis defines radioulnar or preaxial and postaxial orientation, and the DV axis provides dorsal and palmar orientation. Development and differentiation of each axis is controlled by a separate signaling center. The AP axis is defined first, followed by the DV axis, and then the PD axis. The PD axis is controlled by the AER. It secretes WNT3 and fibroblast growth factors (FGFs) including FGF-2, FGF-4, and FGF-8. This provides molecular control for the AER and for the differentiation and limb outgrowth through maintenance of FGF10 expression in the underlying mesoderm. This FGF10 expression sustains proliferation of the PZ mesodermal cells, which in turn are influenced by signaling centers to determine their fate. To maintain PD growth, combined expression of WNT and FGF is required.
AP axis development is controlled by the zone of polarizing activity (ZPA) in the posterior limb mesoderm. The ZPA expands limb width and promotes development of the posterior (ulnar) aspect of the limb through expression of SHH at the distal posterior border of the AER. The AER and ZPA form a reciprocal feedback loop to maintain SHH expression and therefore AP outgrowth.
DV axis growth is regulated by the secretion of WNT7A within the dorsal ectoderm. This induces the Lim homeodomain transcription factor LMX1B, which dorsalizes the underlying limb mesoderm. The ventral ectoderm secretes EN1 and LMX1. WNT7A has also been shown to maintain secretion of SHH (which influences many proteins) from the ZPA, thus linking outgrowth in all three axes.
Signaling centers coordinate control of downstream targets involved in development of the bony skeleton, vessels, muscles, and nerves of the upper limb. These can involve both common and asymmetric molecular pathways. For example, short-stature homeobox 2 (SHOX2) is upregulated in the proximal perichondrium to promote humeral elongation. SHOX, in comparison, is induced in the forearm perichondrium to regulate radioulnar elongation. Appropriate and systematic induction of downstream pathways through coordinated action at multiple signaling centers is essential in normal limb axis differentiation.
32.4 Skeletal Differentiation
At day 32, a hand plate is formed. All bones of the upper limb, except the distal part of the phalanges, form as mesenchymal condensations along the long axis of the limb during week 5. They undergo endochondral ossification in response to growth factors including FGFs, transforming growth factor beta (TGF-β), bone morphogenic proteins (BMP), Indian hedgehog (IHH), retinoids, and parathyroid hormone–related peptide (PTHrP). Chondrification occurs in a proximal to distal direction, and initially results in deposition of cartilage around the entire axis mesenchymal condensation, termed the perichondrium. A cartilaginous model of each bone is created, termed an anlage. Further chondrification is limited to the anlage of each endochondral bone. The mesenchyme in the interzones, which constitutes the sites of future joints, differentiates into fibrous connective tissue. Chondrification of each anlage is followed by ossification. This commences at the primary ossification center. Mesenchymal cells in the perichondrium differentiate into osteoblasts in response to TGF-β and secrete a mineralized bone matrix termed a primary bone collar around the circumference of the bone. This collar thickens as osteoblasts continue to differentiate in progressively more peripheral layers of the perichondrium. Osteoclastic activity facilitates bone remodeling. In the seventh week, the upper limb rotates so that the hands lie anteriorly and the elbows posteriorly.
Formation of the distal portion of the distal phalanges is different, by intramembranous ossification. This means they form directly from condensations of mesenchymal cells without the formation of cartilage intermediary.
Joints develop through repression of chondrogenesis at specific sites of future joints. At these zones, the wingless proteins WTN4 and WTN14 are expressed. The growth factor cartilage-derived morphogenetic protein 1 (CDMP1) is also expressed at these zones to repress chondrogenesis.
By birth, the limb diaphyses are completely ossified, but the epiphyses remain cartilaginous. Secondary ossification centers develop in these epiphyses and ossify. The persisting physis that lies between the epiphysis and metaphysis allows diaphyseal lengthening (growth).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree