Paul H. Bryson OPI Products Inc, Calabasas, CA, USA The use of colored nail polish and nail hardeners has increased among consumers with the rise of the manicure industry. With nail salons found in almost every strip mall, painting nails is a very popular service for the customers of the professional manicurist. The use of nail cosmetics is well‐rooted in history. Ancient Chinese aristocrats colored their nails red or black with polishes made with egg white, beeswax, and gelatin. The Ancient Egyptians used henna to dye the nails a reddish‐brown color (J. Spear, editor of Beauty Launchpad, Creative Age Publications, Van Nuys, CA, personal communication). In the 19th and early 20th centuries, “nail polish” was a colored oil or powder, which was used to rub and buff the nail, literally polishing and coloring the nail simultaneously. Modern nail polish was created in the 1920s, based on early nitrocellulose‐based car paint technology [1]. The term “nail polish” is somewhat of a misnomer for modern products, because no actual polishing is involved in its application. The product is composed of dissolved resins and dries to a hard, glossy coat, so the technically correct name is “nail lacquer.” However, the terms “nail polish,” “nail enamel,” “nail varnish,” “nail paint,” and “nail lacquer” are used interchangeably. Several specialty products have been developed from nail lacquer, including basecoats, topcoats, and hardeners. A newer technology involves pigmented UV‐curable resins. This chapter discusses the current use of these modern formulations (Table 29.1). These nail products are applied by painting the nail with a brush. In best manicuring practices, old nail lacquer is removed with a solvent followed by application of a basecoat, two coats of colored nail lacquer, and a topcoat allowing sufficient time for drying between coats. The basecoat increases the adhesion of the colored nail lacquer to the nail, while the topcoat increases the chip‐resistant characteristics of the colored nail lacquer. These products are applied on both natural and artificial nails. Nail hardener is only applied to natural nails, either as a basecoat or a stand‐alone product. UV‐curing nail “lacquers” are hardened with UV light after application; no evaporation is necessary. In all cases, best practice dictates that the products be kept off the skin. Failure to do so can result in eventual, irreversible sensitization and allergic contact dermatitis [2]. Proper nail cosmetic application dictates the maintenance of excellent hygiene in the nail salon. Unsanitary procedures may result in medical problems [3]. Nail technicians must use cleaned, disinfected, or disposable nail files and tools. Clipping or cutting the cuticles before applying nail lacquer can also lead to infection. Infections with staphylococcus [4] and herpetic whitlows [5] have been attributed to unsanitary manicures. Nail technicians should not perform services on diseased nails. Nail lacquers contain six primary ingredients: resins, solvents, plasticizers, colorants, thixotropic agents, and color stabilizers. By law, all ingredients must be disclosed on the product packaging, using the International Nomenclature for Cosmetic Ingredients (INCI) names. Understanding the chemistry nomenclature is important for isolating the causes of allergic contact dermatitis. Each of these ingredients is discussed in detail (Figures 29.1–29.5). Table 29.1 Overview of product types. Figure 29.1 Lacquered nails. (Source: OPI Products, Inc., Los Angeles, CA. Reproduced with permission.) Figure 29.2 Painting a nail. (Source: OPI Products, Inc., Los Angeles, CA. Reproduced with permission.) Figure 29.3 Be careful with the cuticle. (Source: OPI Products, Inc., Los Angeles, CA. Reproduced with permission.) Figure 29.4 Infected nail. (Source: Nails Magazine. Reproduced with permission.) Resins hold the ingredients of the lacquer together while forming a strong film on the nail. Chemically, the resins are polymers – long‐chain molecules – that are solid or gummy in their pure state. Two types of resins are used. Hard, glossy resins give the lacquered nail its desired appearance; these include nitrocellulose and the methacrylate polymers or co‐polymers (usually labeled by their generic INCI name, “acrylates copolymer”). Topcoat formulations have a higher percentage of these harder resins. Softer, more pliable resins, which enhance adhesion and flexibility, include tosylamide/epoxy resin, tosylamide/formaldehyde resin, and several polyester resins. Basecoats incorporate a higher proportion of pliable resins. Of all the resins, tosylamide/formaldehyde resin is the most commonly implicated in allergic reactions [6] affecting not only the fingers, but other parts of the body by transfer [7]. Solvents are the carriers of the lacquer. They must dissolve the resin, suspend the pigments, and evaporate leaving a smooth film. The drying speed must be controlled to prevent bubbling and skinning; thus faster drying is not necessarily better. Optimum drying speed requires a careful blend of solvents. Ethyl acetate, n‐butyl acetate, and isopropyl alcohol are common solvents; other acetates and alcohols are also occasionally employed. All solvents have a dehydrating and defatting action on the skin, but this usually occurs during the removal of the lacquer, not its application. Formerly, toluene was a commonly used solvent. Research indicates that toluene exposure for a nail technician or consumer was far below safe exposure limits [8]; however, consumer perceptions are negative for toluene, necessitating its replacement. Ketones such as acetone or methyl ethyl ketone (MEK) are not amenable to suspension of pigments and are therefore used at low levels, if at all, in lacquers, although acetone will dissolve the resins effectively and therefore is an effective lacquer remover. A few water‐based nail “lacquers” are now on the market. Because of their slow drying time and lesser durability, they are unlikely to replace solvent‐based products in the foreseeable future. If they are ever perfected, they will completely take over the industry, because water is cheaper, non‐flammable (which reduces shipping costs), and odorless. Figure 29.5 Dermatitis on the finger. (Source: Nails Magazine. Reproduced with permission.) Plasticizers keep the resins flexible and less likely to chip. Camphor and dibutyl phthalate (DBP) have long been used for this purpose; however, the European Union (EU) maintains its 2004 ban of DBP, despite authoritative findings regarding its safety in nail lacquer [9]. Because many manufacturers sell globally, DBP has largely been replaced by other plasticizers, including triphenyl phosphate, trimethyl pentanyl diisobutyrate, acetyl tributyl citrate, ethyl tosylamide, and sucrose benzoate. Colorants are selected from among various internationally accepted pigments. They are mostly used in the “lake” form, meaning that the organic colorants have been adsorbed or co‐precipitated into inorganic, insoluble substrates such as the silicates, oxides, or sulfates of various metals. A shimmer effect is created by minerals such as mica, synthetic mica, or powdered aluminum. Larger glitters are made of polymer flakes. Following INCI convention, most colorant materials are labeled by their international “Color Index” (CI) numbers. This is a convenient way to identify colors, which have different national designations. Labeling colorants by their CI numbers is either legal or de facto accepted by most regulatory agencies around the world; even so, out of deference to local custom, colors are often declared binomially (e.g. CI 77891/Titanium Dioxide) (Figure 29.6). Figure 29.6 Nail lacquer. (Source: OPI Products, Inc., Los Angeles, CA. Reproduced with permission.)
CHAPTER 29
Colored Nail Cosmetics and Hardeners
Introduction
Application techniques
Lacquers, topcoats, and basecoats
Product class
Nail lacquer
Basecoat
Topcoat
Nail hardener
UV curable
Coating created by
Solvent evaporation
Solvent evaporation
Solvent evaporation
Mainly solvent evaporation; some polymerization of formalin may occur
Polymerization
Resin type or mix
Balanced
Biased toward adhesion
Biased towards glossiness, hardness
Balanced or biased towards adhesion
Balanced; resin formed by reacting directly on nail
Pigment
Yes
Little or none
Little or none
Usually none
Yes (except base gel/top gel)
Removal
Easily dissolves in solvent
Easily dissolves in solvent
Easily dissolves in solvent
Easily dissolves in solvent
Soften by acetone soak, then peel
Benefits
Attractive color; can be applied over natural nails or enhancements
Helps color coat last longer; protects natural nail from staining
Helps color coat last longer; some contain optical brighteners or UV protectants
Strengthens natural nail by cross‐linking proteins; may be used as a basecoat
Attractive color; tough cured‐in‐place resin protects nail 

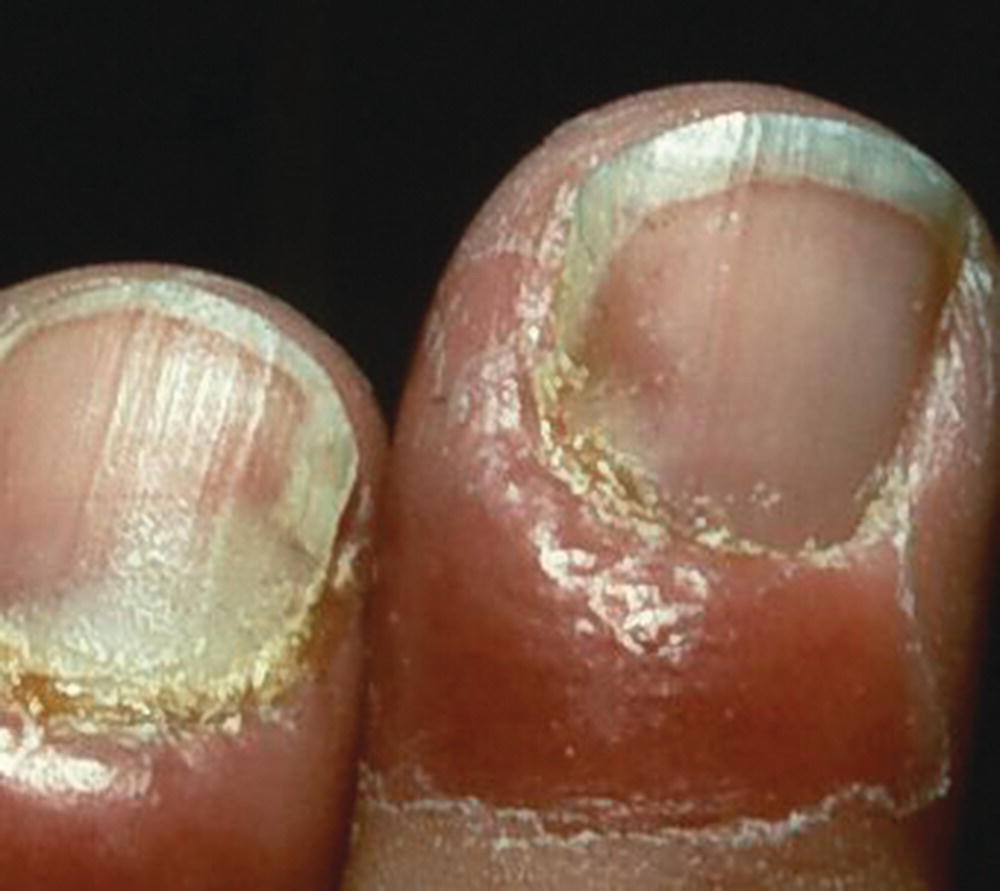
Resins
Solvents
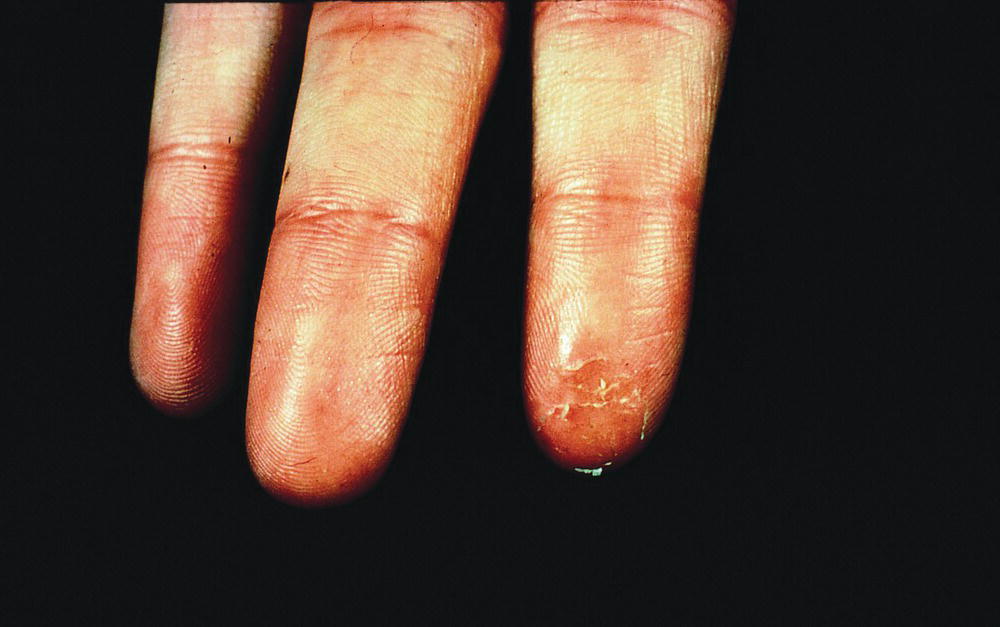
Plasticizers
Colorants

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree