Endocrine, Metabolic and Nutritional Diseases 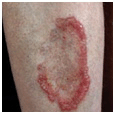
Skin Diseases in Pregnancy
 Normal skin changes associated with pregnancy are darkening of linea alba (linea nigra), melasma (see Section 13), and striae distensae (Fig. 15-1).
Normal skin changes associated with pregnancy are darkening of linea alba (linea nigra), melasma (see Section 13), and striae distensae (Fig. 15-1).
 Pruritus occurring in pregnancy may be due to a flare of preexisting dermatosis or a pregnancy-specific dermatosis.
Pruritus occurring in pregnancy may be due to a flare of preexisting dermatosis or a pregnancy-specific dermatosis.
 Pregnancy-specific dermatoses associated with fetal risk are cholestasis in pregnancy, pustular psoriasis of pregnancy (impetigo herpetiformis), and pemphigoid gestationis.
Pregnancy-specific dermatoses associated with fetal risk are cholestasis in pregnancy, pustular psoriasis of pregnancy (impetigo herpetiformis), and pemphigoid gestationis.
 Pregnancy-specific dermatoses not associated with fetal risk are polymorphic eruption of pregnancy and prurigo gestationis.
Pregnancy-specific dermatoses not associated with fetal risk are polymorphic eruption of pregnancy and prurigo gestationis.
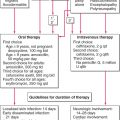
 An algorithm of an approach to a pregnant patient with a pruritus is shown in Fig. 15-2.
An algorithm of an approach to a pregnant patient with a pruritus is shown in Fig. 15-2.
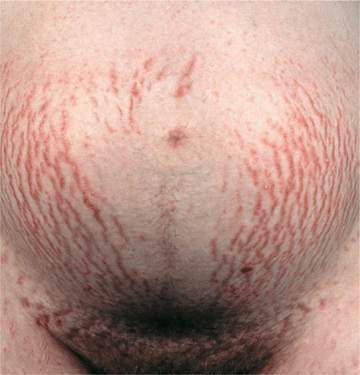
Figure 15-1. Striae distensae in a pregnant woman (36 weeks of gestation).
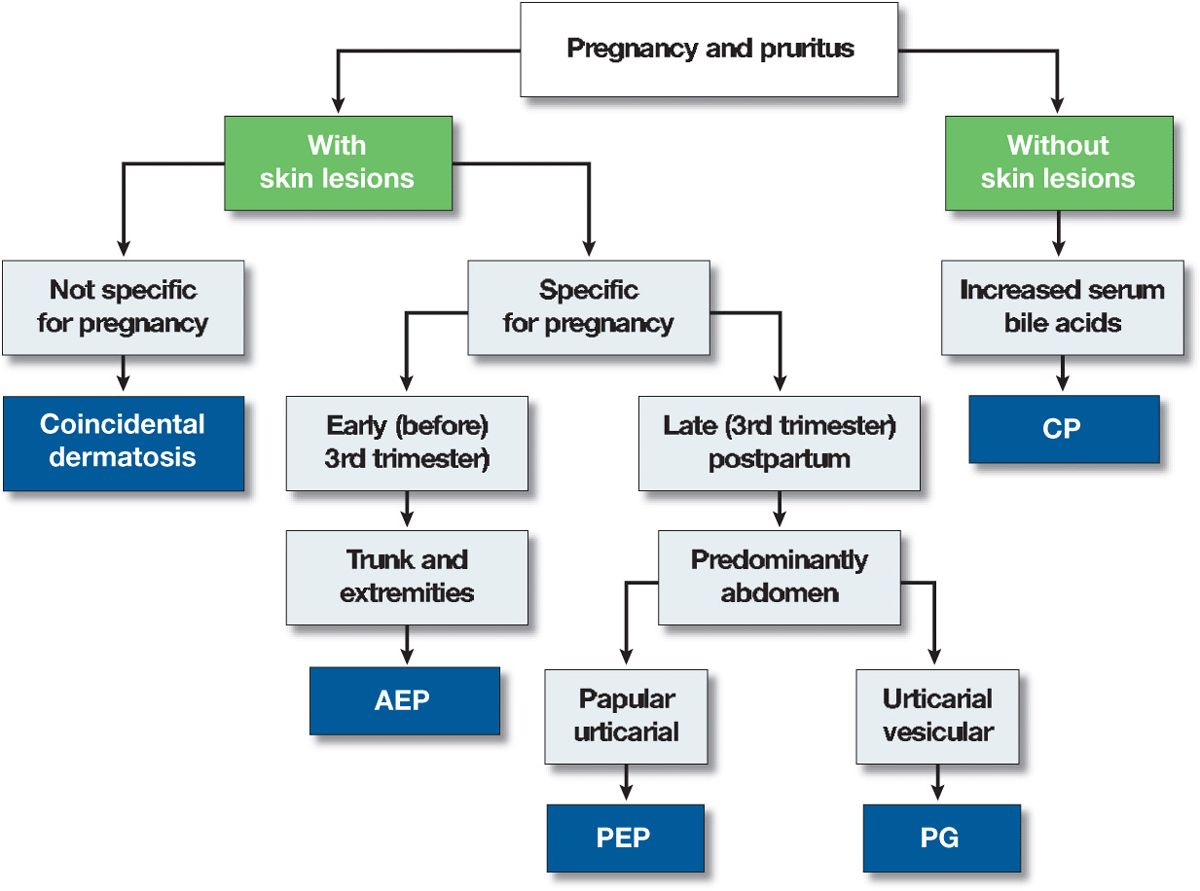
Figure 15-2. Algorithm of approach to a pregnant patient with pruritus. AEP, atopic eruption of pregnancy; PEP, polymorphic eruption of pregnancy; PG, pemphigoid gestationis; CP, cholestasis of pregnancy.
Cholestasis of Pregnancy (CP) ICD-9: 646.7  ICD-10: K83.1
ICD-10: K83.1 
 Occurs in the third trimester.
Occurs in the third trimester.
 Leading symptoms: pruritus, either localized (palms) or generalized. Most severe during the night.
Leading symptoms: pruritus, either localized (palms) or generalized. Most severe during the night.
 Cutaneous lesions invariably absent, but excoriations in severe cases.
Cutaneous lesions invariably absent, but excoriations in severe cases.
 Elevation of serum bile acids.
Elevation of serum bile acids.
 Fetal risks include prematurity, intrapartal distress, and fetal death.
Fetal risks include prematurity, intrapartal distress, and fetal death.
 Treatment: ursodeoxycholic acid, plasmapheresis.
Treatment: ursodeoxycholic acid, plasmapheresis.
Pemphigoid Gestationis ICD-9: 646.8  ICD-10: 026.4
ICD-10: 026.4 
 Pemphigoid gestationis is a pruritic polymorphic inflammatory dermatosis of pregnancy and the postpartum period. It is an autoimmune process with circulating complement-fixing IgG antibodies in the serum. The condition is described in Section 6.
Pemphigoid gestationis is a pruritic polymorphic inflammatory dermatosis of pregnancy and the postpartum period. It is an autoimmune process with circulating complement-fixing IgG antibodies in the serum. The condition is described in Section 6.
Polymorphic Eruption of Pregnancy (PEP) ICD-9: 709.8  ICD-10: 99.740
ICD-10: 99.740 
 PEP is a distinct pruritic eruption of pregnancy that usually begins in the third trimester, most often in primigravidae (76%). Common, estimated to be 1 in 120–240 pregnancies.
PEP is a distinct pruritic eruption of pregnancy that usually begins in the third trimester, most often in primigravidae (76%). Common, estimated to be 1 in 120–240 pregnancies.
 There is no increased risk of fetal morbidity or mortality.
There is no increased risk of fetal morbidity or mortality.
 The etiology and pathogenesis are not understood.
The etiology and pathogenesis are not understood.
 Average time of onset is 36 weeks of gestation, usually 1–2 weeks before delivery. However, symptoms and signs can start in the postpartum period.
Average time of onset is 36 weeks of gestation, usually 1–2 weeks before delivery. However, symptoms and signs can start in the postpartum period.
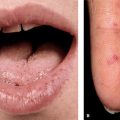
 Severe pruritus develops on the abdomen, often in the striae distensae. Skin lesions consist of erythematous papules, 1–3 mm, quickly coalescing into urticarial plaques (Fig. 15-3) with polycyclic shape and arrangement; blanched halos around the periphery of lesions. Target lesions. Tiny vesicles, 2 mm, but bullae are absent. Although pruritus is the chief symptom, excoriations are infrequent. Affected are the abdomen, buttocks, thighs (Fig. 15-3), upper inner arms, and lower back.
Severe pruritus develops on the abdomen, often in the striae distensae. Skin lesions consist of erythematous papules, 1–3 mm, quickly coalescing into urticarial plaques (Fig. 15-3) with polycyclic shape and arrangement; blanched halos around the periphery of lesions. Target lesions. Tiny vesicles, 2 mm, but bullae are absent. Although pruritus is the chief symptom, excoriations are infrequent. Affected are the abdomen, buttocks, thighs (Fig. 15-3), upper inner arms, and lower back.
 The face, breasts, palms, and soles are rarely involved. The periumbilical area is usually spared. There are no mucous membrane lesions.
The face, breasts, palms, and soles are rarely involved. The periumbilical area is usually spared. There are no mucous membrane lesions.
 Differential diagnosis includes all pruritic abdominal rashes in pregnancy (Fig. 15-2), drug reaction, allergic contact dermatitis, and metabolic pruritus.
Differential diagnosis includes all pruritic abdominal rashes in pregnancy (Fig. 15-2), drug reaction, allergic contact dermatitis, and metabolic pruritus.
 Laboratory findings including histopathology and immunohistopathology are noncontributory.
Laboratory findings including histopathology and immunohistopathology are noncontributory.
 The majority of women do not have a recurrence in the postpartum period, with subsequent pregnancies, or with the use of oral contraceptives. If a recurrence occurs, it is usually much milder.
The majority of women do not have a recurrence in the postpartum period, with subsequent pregnancies, or with the use of oral contraceptives. If a recurrence occurs, it is usually much milder.
 Management: high-potency topical steroids that often can be tapered off, oral prednisone in doses of 10–40 mg/d relieves symptoms in 24 h. Oral antihistamines are ineffective.
Management: high-potency topical steroids that often can be tapered off, oral prednisone in doses of 10–40 mg/d relieves symptoms in 24 h. Oral antihistamines are ineffective.
 Synonyms: PEP, toxemic rash of pregnancy, late-onset prurigo of pregnancy.
Synonyms: PEP, toxemic rash of pregnancy, late-onset prurigo of pregnancy.
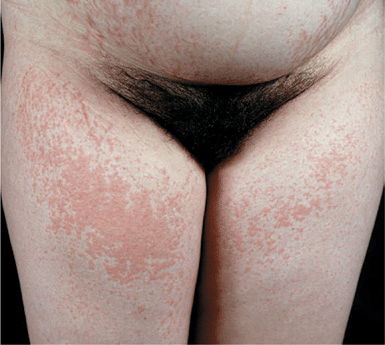
Figure 15-3. Polymorphic eruption of pregnancy [previously called pruritic urticarial papules and plaques of pregnancy (PUPPP)] Urticarial papules are present on both thighs where they coalesce to urticarial plaques. Similar papules and urticarial lesions are present within striae distensae on the abdomen of this pregnant woman at 35 weeks of gestation. Lesions were extremely pruritic, causing sleepless nights and great stress, yet there are no excoriations.
Prurigo of Pregnancy and Atopic Eruption of Pregnancy (AEP) ICD-9: 698–2JJ 782.1 
 Prurigo of pregnancy is now reclassified as part of the AEP spectrum.
Prurigo of pregnancy is now reclassified as part of the AEP spectrum.
 Very common.
Very common.
 AEP consists of flares of atopic dermatitis (also in patients who previously did not have AD); present either with eczematous or prurigo lesions (see Section 2).
AEP consists of flares of atopic dermatitis (also in patients who previously did not have AD); present either with eczematous or prurigo lesions (see Section 2).
 The cardinal symptom is pruritus.
The cardinal symptom is pruritus.
Pustular Psoriasis in Pregnancy ICD-9: 696.7  ICD-10: L40.1
ICD-10: L40.1 
 Previously called impetigo herpetiformis.
Previously called impetigo herpetiformis.
 Clinically and histopathologically indistinguishable from pustular psoriasis of von Zumbusch
Clinically and histopathologically indistinguishable from pustular psoriasis of von Zumbusch
 Burning, smarting, not itching.
Burning, smarting, not itching.
 May have hypocalcemia and decreased vitamin D levels.
May have hypocalcemia and decreased vitamin D levels.
 See “Pustular Psoriasis” in Section 3.
See “Pustular Psoriasis” in Section 3.
Skin Manifestations of Obesity 
 Obesity is widely recognized as an epidemic in the Western world.
Obesity is widely recognized as an epidemic in the Western world.
 Obesity is responsible for changes in skin barrier function, sebaceous glands and sebum production, sweat glands, lymphatics, collagen structure and function, wound healing, micro- and macrocirculation, and subcutaneous fat.
Obesity is responsible for changes in skin barrier function, sebaceous glands and sebum production, sweat glands, lymphatics, collagen structure and function, wound healing, micro- and macrocirculation, and subcutaneous fat.
 Obesity is implicated in a wide spectrum of dermatologic diseases, including acanthosis nigricans (Section 5), acrochordons, keratosis pilaris (Section 4), hyperandrogenlsm and hirsutism (Section 33), striae distensae, adipositas dolorosa and fat redistribution, lymphedema, chronic venous insufficiency, (Section 17) and plantar hyperkeratosis (Section 4).
Obesity is implicated in a wide spectrum of dermatologic diseases, including acanthosis nigricans (Section 5), acrochordons, keratosis pilaris (Section 4), hyperandrogenlsm and hirsutism (Section 33), striae distensae, adipositas dolorosa and fat redistribution, lymphedema, chronic venous insufficiency, (Section 17) and plantar hyperkeratosis (Section 4).
 Cellulitis, skin infections (Section 25), hidradenitis suppurativa (Section 1), psoriasis (Section 3), insulin resistance syndrome, and tophaceous gout (p. 400).
Cellulitis, skin infections (Section 25), hidradenitis suppurativa (Section 1), psoriasis (Section 3), insulin resistance syndrome, and tophaceous gout (p. 400).
Skin Diseases Associated with Diabetes Mellitus*
 Acanthosis nigricans (p. 87) and lipodystrophy.
Acanthosis nigricans (p. 87) and lipodystrophy.
Associated with insulin resistance in diabetes mellitus. Insulin-like epidermal growth factors may cause epidermal hyperplasia.
 Adverse cutaneous drug reactions in diabetes (see Section 23).
Adverse cutaneous drug reactions in diabetes (see Section 23).
Insulin: local reactions—lipodystrophy with decreased adipose tissue at the sites of subcutaneous injection; Arthus-like reaction with urticarial lesion at site of injection.
Systemic insulin allergy: Urticaria, serum sickness–like reactions.
Oral hypoglycemic agents: Exanthematous eruptions, urticaria, erythema multiforme, photosensitivity.
 Calciphylaxis (p. 429)
Calciphylaxis (p. 429)
 Cutaneous perforating disorders
Cutaneous perforating disorders
Rare conditions in which horny plugs perforate into the dermis or dermal debris is eliminated through the epidermis. Not always associated with diabetes (p. 432).
 Diabetic bullae (bullosis diabeticorum) (p. 382).
Diabetic bullae (bullosis diabeticorum) (p. 382).
 Diabetic dermopathy (p. 384).
Diabetic dermopathy (p. 384).
 Eruptive xanthomas (p. 394).
Eruptive xanthomas (p. 394).
 Granuloma annulare (p. 375).
Granuloma annulare (p. 375).
 Infections (see Sections 25 and 26).
Infections (see Sections 25 and 26).
Poorly controlled diabetes associated with increased incidence of primary (furuncles, carbuncles) and secondary Staphylococcus aureus infections (paronychia, wound/ulcer infection), cellulitis (S. aureus, group A streptococcus), erythrasma, dermatophytoses (tinea pedis, onychomycosis), candidiasis (mucosal and cutaneous), mucormycosis with necrotizing nasopharyngeal infections.
 Necrobiosis lipoidica (p. 385).
Necrobiosis lipoidica (p. 385).
 Peripheral neuropathy (diabetic foot) (p. 383).
Peripheral neuropathy (diabetic foot) (p. 383).
 Peripheral vascular disease (see Section 17)
Peripheral vascular disease (see Section 17)
Small-vessel vasculopathy (microangiopathy): Involves arterioles, venules, and capillaries. Characterized by basement membrane thickening and endothelial cell proliferation. Presents clinically as acral erysipelas-like erythema, ± ulceration.
Large-vessel vasculopathy: Incidence greatly increased in diabetes. Ischemia is most often symptomatic on lower legs and feet with gangrene and ulceration. Predisposes to infections.
 Scleredema diabeticorum.
Scleredema diabeticorum.
Synonym: Scleredema adultorum of Buschke. Need not be associated with diabetes. Onset correlates with duration of diabetes and with the presence of microangiopathy. Skin findings: poorly demarcated scleroderma-like induration of the skin and subcutaneous tissue of the upper back, neck, proximal extremities. Rapid onset and progression.
 Scleroderma-like syndrome. Scleroderma-like thickening of skin and limited joint mobility (“prayer sign”).
Scleroderma-like syndrome. Scleroderma-like thickening of skin and limited joint mobility (“prayer sign”).
*Figures in parentheses indicate page numbers where these conditions are dealt with.
Diabetic Bullae ICD-9: 694.8  ICD-10: E14.650
ICD-10: E14.650 
 Large, intact bullae arise spontaneously on the lower legs, feet, dorsa of the hands, and fingers on noninflamed bases (Fig. 15-4).
Large, intact bullae arise spontaneously on the lower legs, feet, dorsa of the hands, and fingers on noninflamed bases (Fig. 15-4).
 When ruptured, oozing bright red erosions result but heal after several weeks.
When ruptured, oozing bright red erosions result but heal after several weeks.
 Localization on dorsa of hand and fingers suggests porphyria cutanea tarda, but abnormalities of porphyrin metabolism are not found.
Localization on dorsa of hand and fingers suggests porphyria cutanea tarda, but abnormalities of porphyrin metabolism are not found.
 Neither trauma nor an immunologic mechanism has been implicated. Histologically, bullae show intra- or subepidermal clefting without acantholysis.
Neither trauma nor an immunologic mechanism has been implicated. Histologically, bullae show intra- or subepidermal clefting without acantholysis.
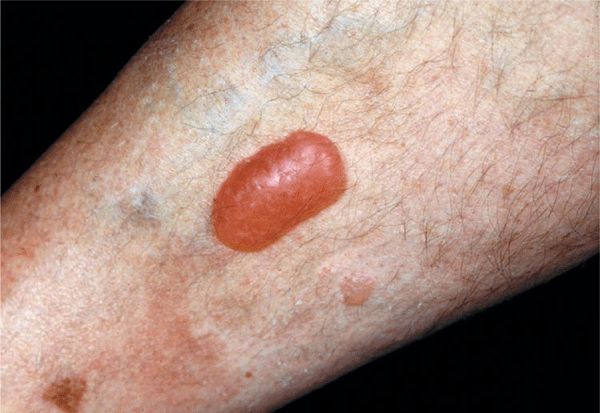
Figure 15-4. Diabetic bulla A large, intact bulla is seen on the pretibial skin on the right lower leg. The patient had many of the vascular complications of diabetes mellitus, i.e., renal failure, retinopathy, and atherosclerosis obliterans resulting in amputation of the left big toe.
“Diabetic Foot” and Diabetic Neuropathy ICD-9: 713.5  ICD-10:G63.2
ICD-10:G63.2
 Peripheral neuropathy is responsible for the “diabetic foot.”
Peripheral neuropathy is responsible for the “diabetic foot.”
 Other factors are angiopathy, atherosclerosis, and infection and most often they are combined.
Other factors are angiopathy, atherosclerosis, and infection and most often they are combined.
 Diabetic neuropathy is combined motor and sensory. Motor neuropathy leads to weakness and muscle wasting distally.
Diabetic neuropathy is combined motor and sensory. Motor neuropathy leads to weakness and muscle wasting distally.
 Autonomic neuropathy accompanies sensory neuropathy and leads to anhidrosis, which may not be confined to the distal extremities.
Autonomic neuropathy accompanies sensory neuropathy and leads to anhidrosis, which may not be confined to the distal extremities.
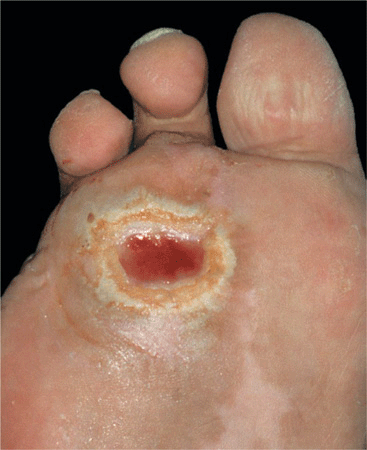
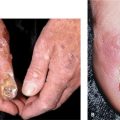
 Sensory neuropathy predisposes to neurotropic ulcers over bony prominences of feet, usually on the great toe and sole (Fig. 15-5).
Sensory neuropathy predisposes to neurotropic ulcers over bony prominences of feet, usually on the great toe and sole (Fig. 15-5).
 Ulcers are surrounded by a ring of callus and may extend to interlying joint and bone, leading to osteomyelitis.
Ulcers are surrounded by a ring of callus and may extend to interlying joint and bone, leading to osteomyelitis.
Figure 15-5. Diabetic, neuropathic ulcer on the sole A large ulcer overlying the second left metacarpophalangeal joint. The patient, a 60-year-old male with diabetes mellitus of 25 years’ duration, has significant sensory neuropathy of the feet and lower legs as well as peripheral vascular disease, which resulted in the amputation of the fourth and fifth toes.
Diabetic Dermopathy ICD-9: 709.8 ° UCD-10: E14:560 
 Circumscribed, atrophic, slightly depressed lesions on the anterior lower legs that are asymptomatic (Fig. 15-6).
Circumscribed, atrophic, slightly depressed lesions on the anterior lower legs that are asymptomatic (Fig. 15-6).
 They arise in crops and gradually resolve, but new lesions appear and occasionally may ulcerate.
They arise in crops and gradually resolve, but new lesions appear and occasionally may ulcerate.
 The pathogenic significance of diabetic dermopathy remains to be established, but it is often accompanied by microangiopathy.
The pathogenic significance of diabetic dermopathy remains to be established, but it is often accompanied by microangiopathy.
Related
Related posts:
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree