Melanoma Precursors and Primary Cutaneous Melanoma 
Epidemiology
Age of Onset. Children and adults.
Prevalence. DN are present in 5% of the general white population. They occur in almost every patient with familial cutaneous melanoma and in 30–50% of patients with sporadic nonfamilial primary melanomas of the skin.
Sex. Equal in males and females.
Race. White persons. Data on persons with brown or black skin are not available; DN are rarely seen in the Japanese population.
Transmission. Autosomal dominant.
Pathogenesis
Multiple loci have been implicated in familial melanoma/DN syndrome, and it is likely that DN is a complex heterogeneous trait. It is assumed that an abnormal clone of melanocytes can be activated by exposure to sunlight. Immunosuppressed patients (renal transplantation) with DN have a higher incidence of melanoma. DN favor the exposed areas of the skin, particularly intermittently sun exposed (e.g., back) and this may be related to the degree of sun exposure; however, DN may also occur in completely covered areas.
Clinical Manifestation
Duration of Lesions. DN usually arise later in childhood than common acquired NMN, which first appear in late childhood, just before puberty. New lesions continue to develop over many years in affected persons; in contrast, common acquired NMN do not appear after middle age and disappear entirely in older persons. DN are thought not to undergo spontaneous regression at all or at least much less than common acquired NMN. Also, whereas common NMN are usually in a roughly comparable stage of development in a given body region (e.g., junctional, compound, dermal), DN appear “out of step,” e.g., a mix of large and small, flat and raised, tan and very dark lesions (Fig. 12-1A).
Skin Symptoms. Asymptomatic.
Family History. In the familial setting, family members can develop melanoma without the presence of DN.
Clinical Features. DN show some of the features of common NMN and some of SSM, so that they occupy an intermediary position between these two morphologies (Table 12-1). No single feature is diagnostic; rather, there is a constellation of findings. They are more irregular, lighter than common NMN, usually maculopapular; have distinct and indistinct borders (Figs. 12-1 and 12-2), and a greater complexity of color than common nevi (Figs. 12-1 and 12-2) but less than melanoma. There are “fried-egg” and “targeted” types (see Fig. 12-20 and Table 12-1). Melanoma arising in a DN appears initially as a small papule (often of a different color) or change in color pattern and massive color change within the precursor lesion (Fig. 12-3).
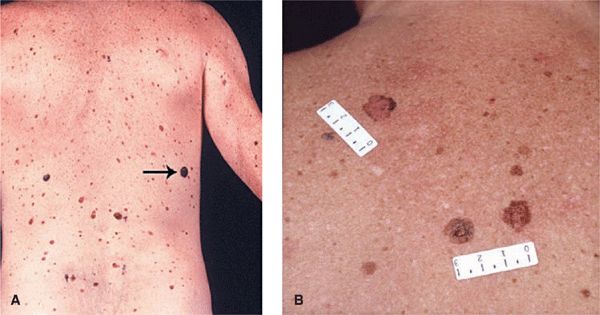
Figure 12-1. Dysplastic nevi (A) Overview of the back of a patient with common and dysplastic nevi. Note a number of lesions are of different size and color, “out of step.” The lesion marked by an arrow was an SSM. (B) Larger magnification of two DNs. Note irregularity and variegation of color that are different in the two lesions (“out of step”). Also, the lesions are 1 cm or larger in diameter. The smaller lesions are common NMN.
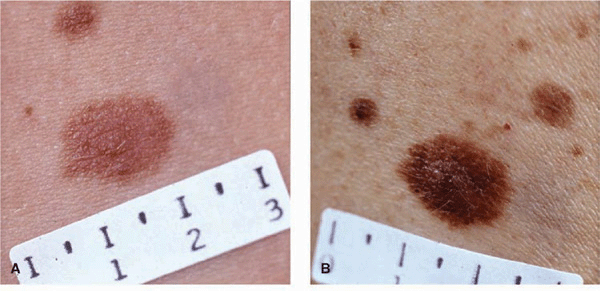
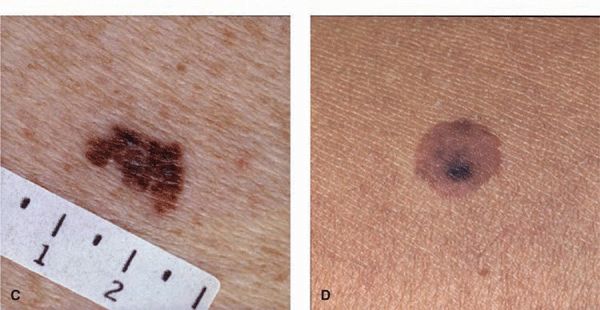
Figure 12-2. Dysplastic nevi (A) A large, uniformly tan, very flat macular oval lesion. The notched border on the left and the size (>1 cm) are the only criteria making this suspicious of a DN. (B) Though relatively symmetric, this lesion is macular and papular with a variegated color and measures 1.5 cm in diameter. The smaller lesions are common NMN. (C) A highly asymmetric, both ill- and sharply defined margin, a notched border, and variegated brown to black color. It is clinically indistinguishable from an SSM (see Figs. 12-12A, B) but was histologically a DN. (D) A relatively symmetric sharply defined lesion with an eccentric, more heavily pigmented area (targetoid lesion).
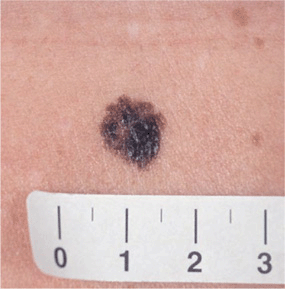
Figure 12-3. Superficial spreading melanoma: arising within a dysplastic nevus The entire lesion originally was maculopapular and had the brown color still seen on the upper crescent-like rim. At a follow-up visit 6 years later, the center and lower half of the lesion had become more raised and turned black as shown here. Melanoma had evolved from a DN. Verified by histopathology.
Table 12-1 COMPARATIVE FEATURES OF COMMON NEVOMELANOCYTIC NEVI (NMN), DYSPLASTIC NEVI (DN), AND SUPERFICIAL SPREADING MELANOMA (SSM)
Dermoscopy. This noninvasive technique allows for clinical improvement of diagnostic accuracy in DN by >50%. Digital dermoscopy permits computerized follow-up of lesions and immediate detection of any change over time, indicating developing malignancy.
Laboratory Examination
Dermatopathology. Hyperplasia and proliferation of melanocytes in a single-file, “lentiginous” pattern in the basal cell layer either as spindle cells or as epithelioid cells and as irregular and dyshesive nests. “Atypical” melanocytes, “bridging” between rete ridges by melanocytic nests; spindle-shaped melanocytes oriented parallel to skin surface. Lamellar fibroplasia and concentric eosinophilic fibrosis (not a constant feature). Histologic atypia do not always correlate with clinical atypia. DN may arise in contiguity with a compound NMN (rarely, a junctional nevus) that is centrally located.
Diagnosis and Differential Diagnosis
The diagnosis of DN is made by clinical recognition of typical distinctive lesions (see Table 12-1), and diagnostic accuracy is considerably improved by dermoscopy. The clinicopathologic correlations are now well documented. Siblings, children, and parents should also be examined for DN once the diagnosis is established in a family member.
Differential Diagnosis. Congenital NMN, common acquired NMN, superficial spreading melanoma, melanoma in situ (MIS), lentigo maligna, Spitz nevus, pigmented basal cell carcinoma.
Association with Melanoma. DN are regarded both as markers for persons at risk for melanoma and as precursors of SSM. Anatomic association (in contiguity) of DN has been observed in 36% of sporadic primary melanomas, in about 70% of familial primary melanomas, and in 94% of melanomas with familial melanoma and DN.
Lifetime Risks of Developing Primary Malignant Melanoma:
• General population: 1.2%.
• Familial DN syndrome with two blood relatives with melanoma: 100%.
• All other patients with DN: 18%.
• The presence of one DN doubles the risk for development of melanoma; with >10 DN, the risk increases 12-fold.
Management
Surgical excision of lesions with narrow margins. Laser or other types of physical destruction should never be used because they do not permit histopathologic verification of diagnosis.
Patients with DN in the familial melanoma setting need to be followed carefully: in familial DN, every 3 months; in sporadic DN, every 6 months to 1 year. Photographic follow-up is important. Most reliable method is digitalized dermoscopy, which should be available in every pigmented lesion and melanoma center. Patients should be given color-illustrated pamphlets that depict the clinical appearance of DN, malignant melanoma, and common acquired NMN. Patients with DN (familial and nonfamilial) should not sunbathe and should use sunscreens when outdoors. They should not use tanning parlors. Family members of the patient should also be examined regularly.
Epidemiology
Prevalence. Present in 1% of white newborns; the majority <3 cm in diameter. Larger CNMN are present in 1:2000 to 1:20,000 newborns. Lesions >9.9 cm in diameter have a prevalence of 1:20,000, and giant CNMN (occupying a major portion of a major anatomic site) occur in 1:500,000 newborns.
Age of Onset. Present at birth (congenital). Some CNMN become visible only after birth (tardive), “fading in” as a relatively large lesion over a period of weeks.
Sex. Equal prevalence in males and females.
Race. All races.
Pathogenesis
Presumably they occur as the result of a developmental defect in neural crest–derived melanoblasts. This defect probably occurs after 10 weeks in utero but before the sixth uterine month; the occurrence of the “split” nevus of the eyelid, i.e., half of the nevus on the upper and half on the lower eyelid, is an indication that nevomelanocytes migrating from the neural crest were in place in this site before the eyelids split (24 weeks).
Small and Large CNMN. CNMN have a rather wide range of clinical features, but the following are typical (Figs. 12-4 and 12-5): CNMN usually distort the skin surface to some degree and are therefore a plaque with or without coarse terminal dark brown or black hairs (hair growth has a delayed onset) (Figs. 12-4B and 12-5B). Sharply demarcated (Fig. 12-4) or merging imperceptibly with surrounding skin; regular or irregular contours. Large lesions may be “wormy” or soft (Fig. 12-5A), rarely firm (desmoplastic type). Skin surface smooth or “pebbly,” mamillated, rugose, cerebriform, bulbous, tuberous, or lobular (Fig. 12-5B). These surface changes are observed more frequently in lesions that extend deep into the reticular dermis.
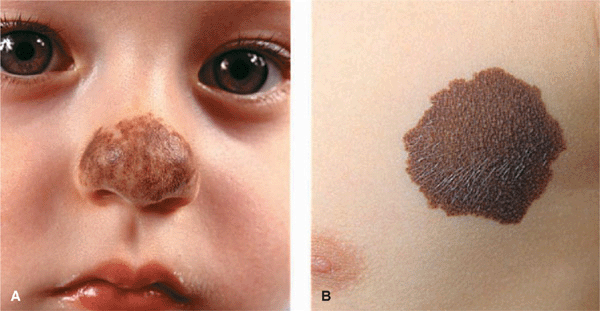
Figure 12-4. Congenital nevomelanocytic nevus (A) Small, variegated brown plaque on the nose. The lesion was present at birth. (B) Congenital nevomelanocytic nevus, intermediate size. Sharply demarcated chocolate-brown plaque with sharply defined borders in an infant. With increasing age, lesions may become elevated and hairy and very discrete hairiness is also noted in this lesion.
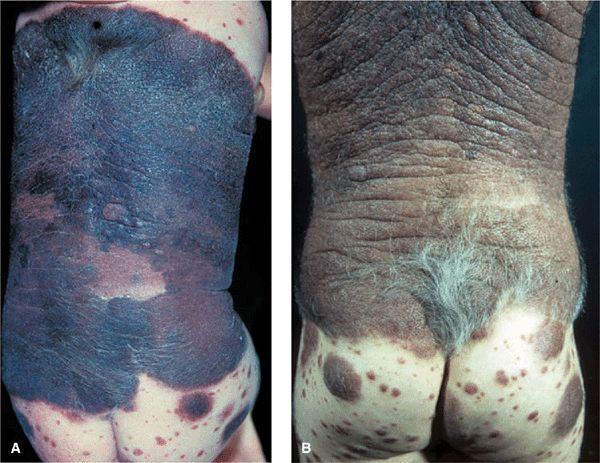
Figure 12-5. A Giant congenital nevomelanocytic nevus (A) In this baby the lesion involves the majority of the skin, with complete replacement of normal skin on the back and multiple smaller CNMN on the buttocks and thighs. There is hypertrichosis in the lower portion. Melanoma developing in a giant CNMN is difficult to diagnose early in a setting of such highly abnormal tissue. (B) Giant CNMN in the same child 5 years later. The CNMN has thickened and has become rugose and more hairy in the sacral region. The lesion is now lighter, i.e. more brown than black and the smaller CNMN on the buttocks have increased in size and number.
Color. Light or dark brown, black. With dermoscopy, a fine speckling of a darker hue with a lighter surrounding brown hue is seen; often the pigmentation is follicular. “Halo” CNMN are rare.
Size. Small (Fig. 12-4), large (>20 cm), or giant (Fig. 12-5). Acquired NMN >1.5 cm in diameter should be regarded as probably tardive CNMN or they represent DN.
Shape. Oval or round.
Distribution of Lesions. Isolated, discrete lesion in any site. Fewer than 5% of CNMN are multiple. Multiple lesions are more common in association with large CNMN. Numerous small CNMN occur in patients with giant CNMN, in whom there may be numerous small CNMN on the trunk and extremities away from the site of the giant CNMN (Fig. 12-5).
Very Large (“Giant”) CNMN
Giant CNMN of the head and neck may be associated with involvement of the leptomeninges with the same pathologic process; this presentation may be asymptomatic or manifested by seizures, focal neurologic defects, or obstructive hydrocephalus. Giant CNMN is usually a plaque with surface distortion, covering entire segments of the trunk, extremities, head, or neck (Fig. 12-5).
Melanoma in CNMN
A papule or nodule arises within CNMN (Fig. 12-6). Often melanoma arises in dermal or subcutaneous nevomelanocytes and can be far advanced when detected.
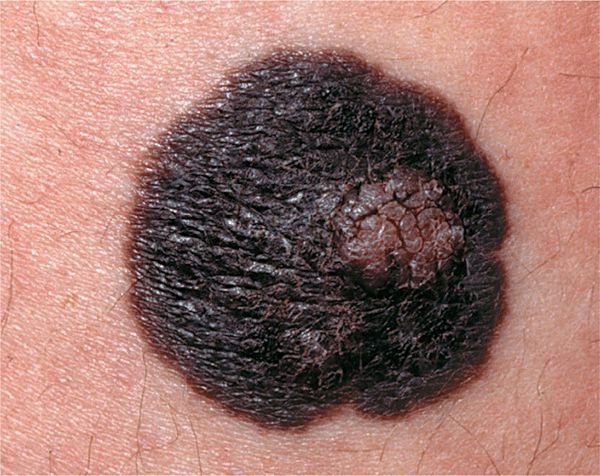
Figure 12-6. Melanoma: arising in small CNMN A black plaque on the thigh of a 36-year-old female, which has been present since birth. Recently, a slightly less pigmented excentric nodule had appeared in this lesion. This is a melanoma.
Differential Diagnosis
Common acquired NMN, DN, congenital blue nevus, nevus spilus, Becker nevus, pigmented epidermal nevi, and café-au-lait macules should be considered in the differential diagnosis of CNMN. Small CNMN are virtually indistinguishable clinically from common acquired NMN except for size, and lesions >1.5 cm may be presumed to be either tardive CNMN or DN.
Laboratory Examination
Histopathology. Nevomelanocytes occur as well-ordered clusters (theques) in the epidermis and in the dermis as sheets, nests, or cords. A diffuse infiltration of strands of nevomelanocytes in the lower one-third of the reticular dermis and subcutis is, when present, quite specific for CNMN. In large and giant CNMN, the nevomelanocytes may extend into the muscle, bone, dura mater, and cranium.
Course and Prognosis
By definition, CNMN appear at birth, but CNMN may arise during infancy (tardive CNMN). The life history of CNMN is not documented, but CNMN can be observed in elderly persons, an age when the common acquired NMN have disappeared.
Large or Giant CNMN. The lifetime risk for development of melanoma in large CNMN has been estimated to be at least 6.3%. In 50% of patients who develop melanoma in large CNMN, the diagnosis is made between the ages of 3 and 5 years. Melanoma that develops in a large CNMN has a poor prognosis because it is detected late.
Small CNMN. The lifetime risk of developing malignant melanoma is 1–5%. The expected association of small CNMN and melanoma is <1:171,000 based on chance alone. Nonetheless, small CNMN should be considered for prophylactic excision at puberty if there are no atypical features (variegated color and irregular borders); small CNMN with atypical features should be excised immediately.
Management
Surgical Excision. The only acceptable method. Small and large CNMN: Excision, with full-thickness skin graft, if required; swing flaps, tissue expanders for large lesions. Giant CNMN: Risk of development of melanoma is significant even in the first 3–5 years of age, and thus giant CNMN should be removed as soon as possible. Individual considerations are necessary (size, location, degree of loss of function, or amount of mutilation). New surgical techniques utilizing the patient’s own normal skin grown in tissue culture can now be used to facilitate removal of very large CNMN. Also, tissue expanders can be used.
Classification of Melanoma
I. De novo melanoma.
A. Melanoma in situ (MIS).
B. Lentigo maligna melanoma (LMM).
C. Superficial spreading melanoma (SSM).
D. Nodular melanoma (NM).
E. Acral lentiginous melanoma (ALM).
F. Melanoma of the mucous membranes.
G. Desmoplastic melanoma.
II. Melanoma arising from precursors.
A. Melanoma arising in dysplastic NMN.
B. Melanoma arising in congenital NMN.
C. Melanoma arising in common NMN.
Four Important Messages Concerning Cutaneous Melanoma
1. Melanoma of the Skin Is Approaching Epidemic Proportions
In 2009, it was estimated that in the United States roughly 122,000 men and women were diagnosed with melanoma of which 69,000 were invasive. Melanoma is a common malignancy and its incidence is on the rise. In the United States, the lifetime risk of invasive melanoma in 2010 was 1 in 50. The US surveillance epidemiology and end results (SEER) estimated 8,650 deaths due to melanoma in the United States. The number of melanomas in the United States continues to increase by 7% per year. Cutaneous melanoma currently represents 5% of newly diagnosed cancer in men and 6% in women. It is the leading fatal illness arising in the skin and is responsible for 80% of deaths from skin cancer. US cancer statistics show that melanoma had the second highest mortality rate increase among men >65 years old. On the other hand, deaths from melanoma occur at a younger age than deaths from most other cancers, and melanoma is among the most common types of cancer in young adults.
2. Early Recognition and Excision of Primary Melanoma Result in Virtual Cure
Current cutaneous melanoma education stresses the detection of early melanoma, with high cure rates after surgical excision. Of all the cancers, melanoma of the skin is the most rewarding for detection of early curable primary tumors, thereby preventing meta-static disease and death. Curability is directly related to size and depth of invasion of the tumor. At present, the most critical tool for conquering this disease is, therefore, the identification of early “thin” melanomas by clinical examination. Total skin examination for melanoma and its precursors should be done routinely.
About 30% of melanomas arise in a preexisting melanocytic lesion; 70% arise in normal skin. Almost all melanomas show an initial radial growth phase followed by a subsequent vertical growth phase. Since metastasis occurs only infrequently during the radial growth phase, detection of early melanomas (i.e., “thin” melanomas) during this phase is essential.
There is the paradox that even with a rising mortality rate, there has been an encouraging improvement in the overall prognosis of melanoma with very high 5-year survival rates (approaching 98%) for thin (<0.75 mm) primary melanoma and an 83% rate for all stages. The favorable prognosis is entirely attributable to early detection.
3. All Physicians and Nurses Have the Responsibility of Detecting Early Melanoma
Early detection of primary melanoma ensures increased survival. The seriousness of this disease thus places the responsibility on the health-care provider in the pivotal role: not to overlook pigmented lesions. Therefore, it is recommended that in clinical practice, no matter what is the presenting complaint, total examination of the body should be requested of all Caucasian patients at the time of the first encounter and that all body regions, including the scalp, toe webs, and orifices (mouth, anus, vulva), be examined.
4. Examination of All Acquired Pigmented Lesions According to the ABCDE Rule
This rule analyzes pigmented lesions according to symmetry, border, color, diameter, growth, and elevation (see p. 261 and Table 12-1). While it does not apply to all types of melanoma, it permits differential diagnostic separation of most melanomas from common nevi and other pigmented lesions.
Etiology and Pathogenesis
The etiology and pathogenesis of cutaneous melanoma are unknown. Epidemiologic studies demonstrate a role for genetic predisposition and sun exposure in melanoma development. The major genes involved in melanoma development reside on chromosome 9p21. Twenty-five to forty percent of members of melanoma-prone families have mutations in cyclin-dependent kinase inhibitor 2A (CDKN2A) and a few families in cyclin-dependent kinase 4 (CDK4). These are tumor-suppressor genes that provide a rational basis for the link to susceptibility to melanoma. Sixty-six percent of melanomas have a mutation of the BRAF gene, others of MC1R.
There is convincing evidence from epidemiologic studies that exposure to solar radiation is the major cause of cutaneous melanoma. Cutaneous melanoma is a greater problem in light-skinned whites (skin types I and II), and sunburns during childhood and intermittent burning exposure in fair skin seem to have a higher impact than cumulative UV exposure over time. Other predisposing and risk factors are the presence of precursor lesions (dysplastic melanocytic nevi and congenital NMN) and a family history of melanoma in parents, children, or siblings. Risk factors for melanoma are listed in Table 12-2.
Table 12-2 RISK FACTORS FOR THE DEVELOPMENT OF MELANOMA
• Genetic markers (CDKN2a), BRAF, MC1R
• Photo skin type I/II
• Family history of dysplastic nevi or melanoma
• Personal history of melanoma
• Ultraviolet irradiation, particularly sunburns during childhood and in termittent burning exposures
• Number (>50) and size (>5 mm) of melanocytic nevi
• Congenital nevi
• Number of dysplastic nevi (>5)
• Dysplastic melanocytic nevus syndrome
Melanoma Growth Patterns
Almost all melanomas show an initial radial growth phase followed by a subsequent vertical growth phase. Radial growth phase refers to a mostly intraepidermal, preinvasive, or minimally invasive growth pattern; vertical growth refers to growth into the dermis and thus into the vicinity of vessels that serve as avenues for metastasis. Since most melanomas produce melanin pigment, even preinvasive melanomas in their radial growth phase are clinically detectable by their color patterns. The prognostic difference among the clinical types of melanoma relates mainly to the duration of the radial growth phase, which may last from years to decades in LMM, from months to 2 years in SSM, and 6 months or less in NM.
Melanoma Recognition
Six Signs of Malignant Melanoma (ABCDE Rule), (does not apply to nodular melanoma)
A. Asymmetry in shape—one-half unlike the other half.
B. Border is irregular—edges irregularly scalloped, notched, sharply defined.
C. Color is not uniform; mottled—haphazard display of colors; all shades of brown, black, gray, blue, red, and white.
D. Diameter is usually large—greater than the tip of a pencil eraser (6.0 mm); others use D for “ugly duckling” sign: lesion is different from other pigmented lesions (nevi) on the body with respect to change in size, shape, and color.
E. Elevation is almost always present and is irregular—surface distortion is assessed by side lighting. MIS and acral lentiginous lesions initially macular; others use E for Evolving. A history of an increase in the size of lesion is one of the most important signs of malignant melanoma.
Clinical Presentations of Melanoma
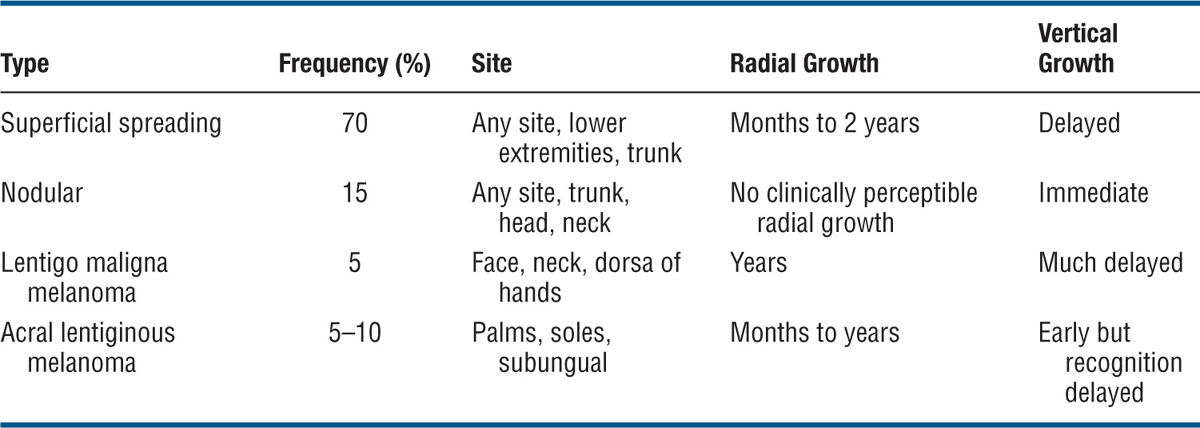
The clinical characteristics of the four major types of melanoma are summarized in Table 12-3. Frequency of melanoma by type of tumor: SSM, 70%; NM, 15%; LMM, 5%; and acral and unclassified melanoma, 10%. Also discussed in this section are MIS and desmoplastic melanoma.
Table 12-3 FOUR MAJOR TYPES OF MELANOMA
 ICD-10: D48–5
ICD-10: D48–5 
 Dysplastic nevi (DN) are a special type of acquired, circumscribed, pigmented lesions that represent disordered proliferations of variably atypical melanocytes.
Dysplastic nevi (DN) are a special type of acquired, circumscribed, pigmented lesions that represent disordered proliferations of variably atypical melanocytes. DN arise de novo or as part of a compound melanocytic nevus.
DN arise de novo or as part of a compound melanocytic nevus. DN are clinically distinctive from common acquired nevi: larger and more variegated in color, asymmetric in outline, irregular borders; they also have characteristic histologic features.
DN are clinically distinctive from common acquired nevi: larger and more variegated in color, asymmetric in outline, irregular borders; they also have characteristic histologic features. DN are regarded as potential precursors of superficial spreading melanoma (SSM) and also as markers of persons at risk for developing primary malignant melanoma of the skin, either within the DN or on normal skin.
DN are regarded as potential precursors of superficial spreading melanoma (SSM) and also as markers of persons at risk for developing primary malignant melanoma of the skin, either within the DN or on normal skin. DN occur either sporadically or in the context of the familial DN syndrome: kindreds with familial multiple DN and melanomas (formerly FAMMM, or B-K mole syndrome).
DN occur either sporadically or in the context of the familial DN syndrome: kindreds with familial multiple DN and melanomas (formerly FAMMM, or B-K mole syndrome). Synonym: atypical melanocytic nevus.
Synonym: atypical melanocytic nevus. ICD-10: D22
ICD-10: D22 
 CNMN are pigmented lesions of the skin usually present at birth; rare varieties of CNMN can develop and become clinically apparent during infancy.
CNMN are pigmented lesions of the skin usually present at birth; rare varieties of CNMN can develop and become clinically apparent during infancy. CNMN may be any size from very small to very large.
CNMN may be any size from very small to very large. CNMN are benign nevomelanocytic neoplasms.
CNMN are benign nevomelanocytic neoplasms. However, all CNMN, regardless of size, may be precursors of malignant melanoma.
However, all CNMN, regardless of size, may be precursors of malignant melanoma. ICD-10: C43
ICD-10: C43 
 Cutaneous melanoma is the most malignant tumor of the skin. Melanoma arises from the malignant transformation of melanocytes at the dermal–epidermal junction or from the nevomelanocytes of DN or CNMN that become invasive and metastasize after various time intervals.
Cutaneous melanoma is the most malignant tumor of the skin. Melanoma arises from the malignant transformation of melanocytes at the dermal–epidermal junction or from the nevomelanocytes of DN or CNMN that become invasive and metastasize after various time intervals. ICD-10: D02
ICD-10: D02 
 The clinical features of MIS are not always clearly presented. MIS is primarily a histopathologic definition, and the term is used when melanoma cells are confined to the epidermis, above the basement membrane; basilar melanocytic atypia, hyperplasia, and spread occur either in single-file alignment along the basal membrane or are distributed throughout the epidermis (pagetoid spread). Every melanoma starts as an in situ lesion, but MIS is clinically diagnosable only when the radial growth phase is long enough for it to become visually detectable. Such lesions are flat, within the level of the skin, and thus a macule (
The clinical features of MIS are not always clearly presented. MIS is primarily a histopathologic definition, and the term is used when melanoma cells are confined to the epidermis, above the basement membrane; basilar melanocytic atypia, hyperplasia, and spread occur either in single-file alignment along the basal membrane or are distributed throughout the epidermis (pagetoid spread). Every melanoma starts as an in situ lesion, but MIS is clinically diagnosable only when the radial growth phase is long enough for it to become visually detectable. Such lesions are flat, within the level of the skin, and thus a macule ( The clinical correlations of MIS are lentigo maligna (
The clinical correlations of MIS are lentigo maligna (