(1)
Dermatologische Praxis & Haarcenter, Wallisellen (Zürich), Switzerland
I think of my body as side effect of my mind.Carrie Fisher (1956–)
A side effect is an effect that is secondary to the one intended. The term is primarily used to describe unwanted or adverse effects. In the broader sense, the term can also apply to unintended, but beneficial effects from the use of the drug. An example is the hair growth-promoting effect of minoxidil that was originally a drug intended to lower the blood pressure but proved to be a powerful trichotrophic agent.
Occurrence or fear of adverse effects is a major barrier to patient compliance. Therefore, it is of upmost importance to inform patients on potential adverse effects, their frequencies, and appropriate management.
Some adverse effects are directly related to the pharmacological effect of the drug and others to a patient’s individual allergic or idiosyncratic disposition, to drug–drug interactions, or to simply not following instructions for proper use of the medication. Patients should be aware of possible allergies to active ingredients or additives, and report other medical conditions and medications. Therefore, it is highly recommended, even when treatment can be purchased without a doctor’s prescription, that the patient visits a physician for proper indication, exclusion of contraindications, instructions for proper use, and follow-up.
Topical minoxidil and oral finasteride are the most frequently used drugs for treatment of alopecia. For a comprehensive list of reported adverse reactions to other drugs used for treatment of hair loss and related disorders, the readers are encouraged to refer to the respective information for consumers and healthcare professionals accompanying the drug.
6.1 Adverse Reactions to Topical Minoxidil
Topical minoxidil solution and foam are used to promote hair growth in the treatment of androgenetic alopecia. Minoxidil is not indicated for treatment of telogen effluvium or alopecia areata, lest androgenetic alopecia represents a comorbidity. Efficacy and safety data are not available before the age of 18 years and after the age of 65 years. Nevertheless, minoxidil can be safely used before 18 or after 65 years at the discretion of the prescriber (off-label use).
Children are at higher risk of adverse cardiovascular effects of topical minoxidil solution.
Therefore, before the age of 12 years, the dosage should be halved. Any of the following health problems should caution to the use of topical minoxidil: active diseases of the scalp, e.g., eczema, infection, and cuts, as well as heart problems, e.g., chest pain, heart attack, and heart failure.
Before applying minoxidil to the scalp, the area should be clean and dry. The product may be applied to damp hair. To use the solution, the applicator is filled with 1 ml of medication. The hair in the area of thinning is parted, and the solution is applied evenly to the affected area of the scalp and gently rubbed in. The solution or foam is applied twice a day. The solution is allowed to dry completely before using other styling products, e.g., gels or mousse, or before going to bed. If a dose is missed, the missed dose is to be skipped and the usual dosing schedule resumed. The dose should not be doubled to catch up.
Upon initiation of treatment with topical minoxidil, patients may experience temporary increased shedding of hair (shedding phase). Patients should be prepared and informed that this represents a physiological response to treatment, since minoxidil not only increases the duration of anagen in the hair cycle but also triggers an immediate telogen release.
The best thing to do is to continue treatment until the shedding stops (usually within 6 weeks) and new hair growth is seen (usually at 3 months). Patients should also be aware that it takes time for hair to regrow. Most people need to use this medication regularly for at least 8 weeks to see a benefit. If the condition does not improve or worsens after using this medication for 3–6 months, the condition and treatment need to be reevaluated by the physician. The effect of minoxidil builds up until 12 months of treatment. Once it is established that minoxidil is effective, treatment should thereafter continue indefinitely since positive results will be reversed within 6 weeks to 3 months once treatment is stopped.
Another common adverse effect of minoxidil is elongation, thickening, and enhanced pigmentation of fine facial hair and rarely of body hair (hypertrichosis). This develops relatively early in the course of treatment (usually within 6 weeks after starting therapy). It is usually first noticed on the temples (Fig. 6.1a.), between the hairline and the eyebrows, or in the sideburn area of the upper lateral cheek (Fig. 6.1b). Upon discontinuation of minoxidil, new hair growth stops, but up to 12 months may be required for restoration to pretreatment appearance.
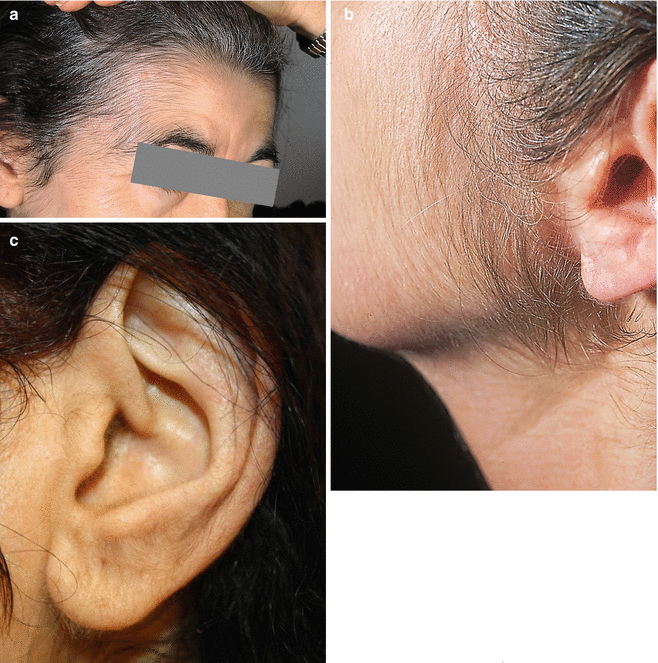

Fig. 6.1
(a–c) Minoxidil-induced hypertrichosis. (a) On the temple. (b) Of the sideburn area and lateral cheek. (c) Of the ear
Minoxidil-induced hair growth may be especially disturbing to women. Patients should therefore be carefully informed about this possible effect before treatment is started.
The risk is significantly higher with 5 % topical minoxidil, in women with preexistent hypertrichosis, especially those of Mediterranean or Indian origin. Disturbing hairs may be bleached, clipped, or waxed.
Itching, redness, or irritation at the treated area are far more frequently due to an irritant dermatitis to propylene glycol or the alcohol-based solution of minoxidil.
If irritant scalp dermatitis is a problem, either minoxidil foam or a minoxidil compound should be used that are free of propylene glycol.
Rp. | |
|---|---|
Minoxidil | 2.0, 3.0, or 5.0 g |
Glycerin | 10.0 g |
Water | 20.0 g |
Ethanol 96 % | ad 100.0 g |
M(isce).D(a).S(igna). Apply twice daily 1 ml to the affected area | |
After applying minoxidil onto the scalp, exposure to the sun should be avoided, since it may cause sunburns. Minoxidil should not be used on skin that is red, painful, irritated, scraped, cut, or infected, since doing so may cause the drug to be absorbed into the body and result in systemic side effects. Hands should be thoroughly washed after application. Caution is to be given to avoid getting the medication in eyes. If this occurs, eyes should be rinsed with large amounts of cool water.
Very few minoxidil users are truly allergic to the active ingredient. While irritant dermatitis usually develops early in the course of treatment, allergic reactions are usually acquired later.
If an allergic reaction to minoxidil is suspected (Fig. 6.2a), this can be clarified by performing a repeated open application test (ROAT) to the forearm: minoxidil is applied twice daily to an area of 5 × 5 cm on the inner aspect of the forearm for a duration of 7 days. In the event of a contact allergic reaction, an erythematous rash with papules will become apparent usually within 48–72 h (Fig. 6.2b).
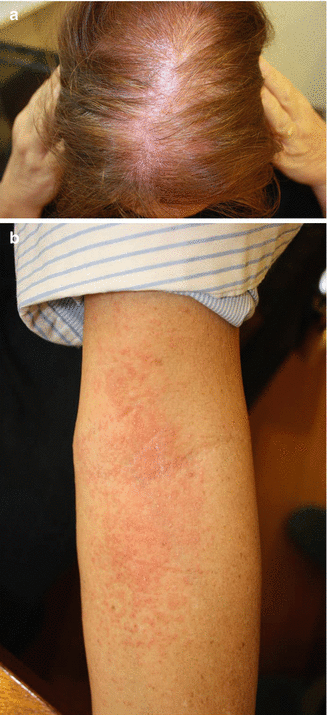

Fig. 6.2
(a, b) Allergic contact dermatitis to minoxidil. (a) Of the application area and (b) positive ROAT
While most of patients with allergic contact dermatitis described in the literature showed a positive sensitization to the vehicle substance propylene glycol evaluated by patch testing, reactions to the active ingredient minoxidil are rare. Hagemann et al. reported a case of allergic sensitization to minoxidil, which they evaluated and differentiated from an irritative reaction by a combination of patch testing and lymphocyte transformation test (LTT).
The differentiation of allergic and irritative adverse effects of topical minoxidil solutions and the identification of the causative agent are of major relevance for the proceeding and adjustment of therapy.
Patients with sensitizations against propylene glycol are candidates for preparations with alternative solvents but can proceed treatment with minoxidil. In contrast, patients with allergies to the active ingredient itself are no longer candidates for treatment with minoxidil and should undergo alternative therapeutic options.
Other reported adverse reactions to topical minoxidil solution include migraine headache, erosive pustular dermatosis of the scalp, pustular allergic contact dermatitis, pigmented contact dermatitis, leukoderma, central chorioretinopathy, severe hypertrichosis of the external ear canal, and trichostasis spinulosa of the forehead (Fig. 6.3). Adverse reactions to topical minoxidil are summarized in Table. 6.1.
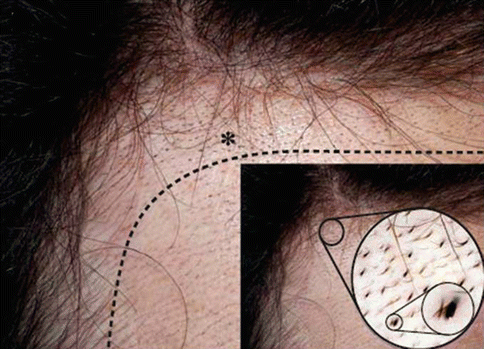

Fig. 6.3
Minoxidil-induced trichostasis spinulosa. The temporal region shows multiple small blackish spines extruding from hair follicle openings. The dotted line indicates the approximate area of minoxidil application. The asterisk indicates the region evaluated by dermoscopy shown in the inset at original magnification × 10 (From Navarin A, Ziegler M, Kolm I et al (2010) Arch Dermatol 146:1434–1435)
Table 6.1
Adverse reactions to 2 or 5 % topical minoxidil solution
Common: |
Hair shedding (during initial 4–6 weeks of treatment) |
Unwanted hair growth elsewhere on the body (usually facial in women, within 3–6 weeks of treatment: Fig. 6.2) |
Itching, redness, or irritation at the treated area (usually due to propylene glycol) |
Changes in hair color or texture |
Burning or irritation of the eye |
Rare (treatment should be withheld immediately): |
Severe allergic reactions: rash, hives, itching, difficulty breathing, tightness in the chest, swelling of the mouth, face, lips, or tongue |
Cardiovascular effects: chest pain; dizziness; fainting; increased heart rate; pounding heartbeat; sudden, unexplained weight gain; swollen hands or feet |
Unexplained: breast tenderness, changes in vision or hearing |
Thrombocytopenia and leukopenia (WBC < 3,000/mm3) and toxic epidermal necrolysis (TEN) have very rarely been reported with oral minoxidil |
Cases of fetal malformation (brain, heart, and vascular), of caudal regression syndrome, and of hypertrichosis of the infant were reported from the use of topical minoxidil during pregnancy.
Eight deaths occurred during the original Upjohn-sponsored clinical trials with topical minoxidil and 2 deaths in subjects who used extemporaneous formulations of the drug. Of the 8 patients, 5 had cardiovascular abnormalities, 2 had acquired immunodeficiency syndrome-related pneumonia, and one patient died of a self-inflicted gunshot wound. Of the subjects who were using extemporaneous topical minoxidil, one had hypertension and arteriosclerotic disease, and the other died of myocardial infarction.
There is a little likelihood of serious adverse effects from the proper use of topical minoxidil in otherwise healthy, adult patients, and the evidence suggests that reported deaths were the result of causes other than the use of minoxidil because of its low systemic absorption.
The percutaneous absorption and excretion of 5 % solution of minoxidil labeled with carbon 14 were measured in adult male subjects, who received nine topical applications to a bald area on the scalp, with the radioactive solutions applied on days 1 and 9. Urinary excretion of radioactivity was low, with mean values ranging from 1.6 to 3.9 % of applied dose. No radioactivity was detected in fecal samples. Recovery of radioactivity from the skin surface and from scalp and pillowcase washes was in the range of 41–45 % of applied dose.
Although minoxidil is poorly absorbed through the skin, systemic doses in the range of 2.4–5.4 mg/day can be anticipated if application is made to the entire scalp. Therefore, it is recommended to refrain from the use of topical minoxidil in patients with significant and arrhythmogenic cardiovascular disease.
Healthy male volunteers completed a four-way, multiple-dose, randomized crossover study to determine the relationship between contact time of applied drug on the scalp and minoxidil absorption from a 2 % topical solution. One milliliter of solution was applied twice daily over 150 cm2 of bald scalp to each subject for 6 days. Unabsorbed drug was washed off the scalp after 1, 2, 4, and 11.5 h of contact time in each of four treatments. Cumulative urinary excretion profiles within steady-state, 12-h dosing intervals were well described by straight lines for all treatments, indicating that systemic minoxidil elimination was rate controlled by constant, zero-order percutaneous drug absorption. The extent of minoxidil absorption, expressed as steady-state urinary excretion of unchanged minoxidil, minoxidil glucuronide, or the sum of these, increased in a disproportionate manner with increase in contact time of drug on the scalp.
Relative to the amount of topical minoxidil absorbed after a contact time of 11.5 h, absorption was approximately 50 % complete by 1 h and greater than 75 % complete by 4 h.
This suggests that minoxidil absorption from the vehicle into skin occurs rapidly relative to diffusion through skin. The rate of minoxidil absorption from vehicle into skin was characterized as nonlinear, whereas minoxidil excretion into urine was rate controlled by diffusion from one or more components of the skin which apparently serve as a reservoir, or depot, for minoxidil.
The effect of application frequency on the systemic absorption of topical minoxidil was studied in 52 normal men. Subjects received 1 ml 3 % minoxidil solution applied four, six, or eight times daily to the scalp. Serum and urine were collected and analyzed for minoxidil. No systemic minoxidil accumulation occurred from increasing application frequency to the scalp.
Systemic minoxidil accumulation resulting from frequent application is unlikely. The initial dose probably saturates the skin for a period of time longer than the dosing intervals examined.
Since the efficacy of combining topical 5 % minoxidil with 0.01 % tretinoin once daily was demonstrated to be equivalent to that of conventional 5 % topical minoxidil solution twice daily, a combination of topical minoxidil with tretinoin in a compound has been suggested for treatment of androgenetic alopecia.
However, healthy male volunteers completed a three-way, randomized, crossover study to determine the effect of tretinoin on percutaneous absorption of minoxidil. Subjects received, for 20 days, twice-daily administrations of 1 ml aqueous 2 % topical minoxidil solution either alone, with once-daily applications of a 0.05 % tretinoin cream, or with once-daily applications of a vehicle control cream.
When minoxidil was co-administered with tretinoin, minoxidil absorption was increased nearly threefold, increasing the risk of systemic adverse effects.
Transepidermal water loss measurements, which are sensitive to changes in stratum corneum function, were also significantly increased with tretinoin. No treatment-related changes in stratum corneum thickness were observed on the basis of skin biopsy analysis. The findings indicate that percutaneous minoxidil absorption is enhanced by tretinoin as a result of increased stratum corneum permeability.
Moreover, the combination of topical minoxidil with tretinoin enhances the risk of irritant contact dermatitis of the scalp.
6.2 Adverse Reactions to Oral Finasteride
One milligram oral finasteride received FDA approval for treatment of male pattern baldness in 1997. In several clinical studies, oral finasteride, like topical minoxidil solution, was shown to work on both the crown area and the hairline, but is most successful in the crown area. Again, finasteride is effective only for as long as it is taken. The hair gained or maintained is lost within 6–12 months of stopping treatment.
In a more recent 10-year study of 118 men between 20 and 61 years of age treated with 1 mg oral finasteride daily for androgenic alopecia, Rossi et al. found that 86 % of men continued to benefit from treatment over the entire course of 10 years. In the opinion of the authors, the result after the first year could help in predicting the effectiveness of the treatment; however, many of those unchanged after 1 year of treatment were found to improve later on. Interestingly, better improvements were observed in patients older than 30 years with Hamilton–Norwood grades IV and V. Treatment efficacy was not reduced over time (including those over 40 years of age). Adverse effects were reported in 5.9 % of patients; however, some of them went on with treatment because of the good results.
In the most recent study published in 2012, Sato and Takeda reported on efficacy and safety of 1 mg oral finasteride for treatment of male androgenetic alopecia in the so far largest population study. The study enrolled 3,177 Japanese men treated with oral finasteride from January 2006 to June 2009 at the Tokyo Memorial Clinic Hirayama Department of Plastic and Aesthetic Surgery, School of Medicine, Kitasato University, Tokyo, Japan. Efficacy was evaluated by global photographic assessment, and safety data were assessed by interviews and laboratory tests. The overall effect on hair growth was seen in 87.1 %, in whom hair increased greatly in 11.1 %, moderately in 36.5 %, and slightly in 39.5 %. The response rate improved with increasing duration of treatment. Adverse reactions occurred in 0.7 % of men. Seven men discontinued treatment based on risk–benefit considerations. No specific safety problems associated with long-term use were observed. The authors concluded that in Japanese men with androgenetic alopecia, 1 mg oral finasteride used for long-term treatment maintains progressive hair regrowth without recognized side effect.
Nevertheless, since the original report of persistent diminished libido, erectile dysfunction, and depression among former users of oral finasteride by Traish et al. in 2011, there have been several reports on sexual side effects that may persist despite discontinuation of finasteride medication. These subjects also reported a wide range of symptoms including changes in cognition, ejaculate quality, and genital sensations. Ultimately, Irwig reported depressive symptoms and suicidal thoughts among former users of finasteride with persistent sexual side effects. The author concluded that prescribers of finasteride and men contemplating its use should be made aware of these potential adverse medication effects.
With respect to finasteride and male fertility, early studies did not show any significant influence of finasteride on parameters of semen quality, whereas some later case reports suggested that in subfertile patients, the effects of the drug might be amplified. Therefore, respective counseling may be particularly challenging for male partners of couples who are attempting to become pregnant.
Some users, in an effort to save money, buy 5 mg finasteride tablets instead of the original 1 mg pill and split them into several parts to approximate the 1 mg dosage. Also, finasteride 1 mg capsules formulated with an active ingredient of unknown origin are marketed unofficially to treat androgenetic alopecia in men. Navarro Guerrero et al. performed a galenic-pharmaceutical study of pieces of Proscar® (5 mg finasteride) tablets and finasteride 1 mg capsules versus Propecia® in the treatment of androgenetic alopecia, with the effort to ascertain the pharmaceutical quality of these alternative preparations. The results obtained demonstrated a high variability in the dosage of pieces of tablets ranging from 0.49 mg to 1.81 mg of finasteride, only 10 % being within the dosage range, which is ±5 %, as compared with the authorized 1 mg dose. Applying the criteria of the mass uniformity test, set down in the European Pharmacopoeia, to the set of Proscar® pieces and to the value of the standard deviation percentage (18 %) obtained, the batch would have to be rejected. The finasteride 1 mg capsules showed variable in vitro availability values between 51 and 86 % as compared to Propecia®, which has 92 % availability. Some of these formulations again do not meet the availability specifications established by standard pharmacopeias. Furthermore, the dissolution efficiency of the capsules (from 27 to 76 %) was significantly lower than that of the reference proprietary drug with 82 % (unpublished data).
Finally, the 1 mg finasteride pills are coated to prevent contact with the active ingredient during handling, and the dust or crumbs from broken 5 mg finasteride tablets should be kept away from pregnant women or women who may become pregnant due to teratogenicity.
Splitting 5 mg finasteride tablets to treat androgenetic alopecia is an inappropriate practice from the point of view of safety and dosage variability. With regard to the finasteride 1 mg capsules marketed unofficially, any finasteride formulation purportedly similar to the reference proprietary drug must prove its bioequivalence or interchangeability with respect to the availability of the active ingredient.
Due to interference with doping assays, many sports organizations had banned oral finasteride because it could be misused to mask steroid abuse. Not until 2009, finasteride was removed from the World Anti-Doping Agency’s list of banned substances.
The Prostate Cancer Prevention Trial (PCPT) was a landmark study that sought to reduce the morbidity and mortality of prostate cancer with the use of oral finasteride to prevent disease. The investigators randomly assigned 18,882 men 55 years of age or older with a normal digital rectal examination and a prostate-specific antigen (PSA) level of 3.0 ng per milliliter or lower to treatment with finasteride (5 mg per day) or placebo for 7 years. Prostate biopsy was performed if the annual PSA level, adjusted for the effect of finasteride, exceeded 4.0 ng per milliliter or if the digital rectal examination was abnormal. Prostate cancer was detected in 803 of the 4,368 men in the finasteride group who had data for the final analysis (18.4 %) and 1,147 of the 4,692 men in the placebo group who had such data (24.4 %), for a 24.8 % reduction in prevalence over the 7-year period (95 % confidence interval, 18.6–30.6 %; p < 0.001). Tumors of Gleason grade 7 or more were more common in the finasteride group (280 of 757 tumors [37.0 %] or 6.4 % of the 4,368 men included in the final analysis) than in the placebo group (237 of 1,068 tumors [22.2 %], p < 0.001 for the comparison between groups, or 5.1 % of the 4,692 men included in the final analysis, p = 0.005 for the comparison between groups). Sexual side effects were more common in finasteride-treated men, whereas urinary symptoms were more common in men receiving placebo.
To seek to alleviate the concern of the increase of number of high-grade cancers detected in the original PCPT, Thompson et al. examined long-term all-cause mortality among PCPT participants. The results were reassuring with an overall 15-year rate of death of 22 % in both the finasteride group and the control group. The investigators were unable to report prostate-cancer-specific mortality.Adverse effects to oral finasteride are listed in Table 6.2.
Table 6.2
Adverse reactions to 1 mg oral finasteride
Common (frequency between ≥1/100 and 1/10): |
Sexual dysfunction (finasteride 3.8 % vs. 2.1 % within first 12 months of treatment; 1 % of men withdrew finasteride because of sexual dysfunction within first 12 months of treatment; thereafter, frequency decreased to 0.6 % during following 4 years of treatment) |
Diminished libido (finasteride 1.8 % vs. placebo 1.3 %) |
Erectile dysfunction (finasteride 1.3% vs. placebo 0.7%) |
Occasional (frequency between (≥1/1,000 and <1/100): |
Abnormal ejaculation |
Decreased ejaculatory volume |
Rare (frequency between ≥1/10,000 and <1/1,000): |
Testicular pain |
Breast tenderness |
Gynecomastia (may persist for months to years after cessation of finasteride treatment) |
Allergic reactions: rash, itching, hives, swelling of the mouth, face, lips, or tongue (angioedema) |
Very rare (frequency <1/10,000): |
Depression |
Male breast cancer |
Unknown (frequency cannot be estimated from existing data): |
Persistent diminished libido or erectile dysfunction (after cessation of finasteride treatment) |
Male infertility (usually in association with preexistent subfertility) |
Decrease in quality of semen (there are reports on normalization or improvement of semen quality following withdrawal of drug) |
Post-finasteride syndrome |
Cause-relationship unresolved: |
High Gleason grade tumor (7–10) in the event of prostate carcinoma in men >55 years treated with 5 mg oral finasteride daily for benign prostatic hyperplasia (finasteride 6.4 % vs. placebo 5.1 %) |
In men 55 years or older, 5 mg oral finasteride prevents or delays the appearance of prostate cancer, but this possible benefit and a reduced risk of urinary problems must be weighed against sexual side effects. The increased risk of high-grade prostate cancer detected with finasteride in the original Prostate Cancer Prevention Trial did not prove to affect the overall 15-year rate of death in both the finasteride and the control group in a subsequent study, and the investigators were unable to report prostate-cancer-specific mortality.
6.3 Post-Finasteride Syndrome
The post-finasteride syndrome has been claimed to occur in men who have taken oral finasteride to treat either hair loss or benign prostatic hyperplasia. Reported symptoms include loss of libido, erectile dysfunction, reduction in penis size, penile curvature or reduced sensation, gynecomastia, muscle atrophy, cognitive impairment, severely dry skin, and depression. The condition allegedly may have a life-altering impact on sufferers and their families, such as job loss and the breakup of romantic relationships or marriages while also being linked to suicides.
As yet, the condition is not recognized by the scientific community, although individuals who suffer from the syndrome do present with very distinctive and relatively homogenous symptoms.
While the incidence of persistent sexual, mental, and physical side effects which continue despite quitting finasteride is unknown, it is likely that over 1,000 men worldwide are experiencing the effects. This estimate is based on the number of registered users on the Internet forum (www.propeciahelp.com).
Persistent neurological effects from other drugs are well recognized, such as the tardive dyskinesias related to the use of phenothiazines for treatment of chronic schizophrenia. There is a growing body of scientific evidence from studies in rodents that finasteride may reduce the concentration of several neuroactive steroids important for neurogenesis and neuronal survival. An important neurosteroid is allopregnanolone (ALLO), a metabolite of dihydroprogesterone. ALLO is a potent ligand of the inhibitory GABA–barbiturate receptor. GABAA receptors have variable sensitivities to ALLO in the settings of neurosteroid withdrawal, stress, social isolation, and aging. Less ALLO, as a consequence of finasteride treatment, could alter GABAergic transmission with implications for neuronal progenitors and young neurons. Interestingly, the mechanisms of irreversible tardive dyskinesias from phenothiazines may be similar to the mechanisms underlying the persistent side effects of finasteride: in rats treated with haloperidol to induce orofacial dyskinesias, co-administration of progesterone prevented this side effect, while pretreatment of the rats with finasteride reversed this protective effect, demonstrating an important role of the progesterone pathway and its metabolites.
The Post-Finasteride Syndrome Foundation (www.pfsfoundation.org) is a nonprofit organization dedicated to helping fund research on the characterization, underlying biologic mechanisms, and treatments of the post-finasteride syndrome while improving public awareness of the condition.
Up to date there are no predictive factors for the risk of development of post-finasteride syndrome and no known treatment for the disorder. For the time being, as a general rule:
Refrain from prescribing oral finasteride to a patient with a personal history of depression, sexual dysfunction, or fertility problems.
When fertility is an issue, one may consider performing a sperm count before and during treatment with oral finasteride.
In any case of adverse effects, stop oral finasteride treatment.
In all men 45 and over, perform PSA before and after starting therapy with oral finasteride and thereafter on twice-yearly basis. The level should drop by ca. 50 % upon initiation of therapy. In case of increase >0.4 ng/ml per year, refer to urologist to check prostate condition.
For men who choose regular prostate-cancer screening, the use of oral finasteride meaningfully reduces the risk of prostate cancer.
6.4 Adverse Reactions from Hair Transplantation Surgery
Autologous hair transplantation remains the only treatment option for advanced patterned baldness both in males and in females and may be performed in selected cases of cicatricial alopecia. With the advent of total micrografting and a sharp decline in scalp reduction procedures, hair restoration surgery has become more satisfying than in the recent past. Modern techniques use the visual qualities of hair and varying design patterns to create the optical illusion of more hair than is actually present. The main advances in hair restoration surgery have been the use of one to four hair grafts, which allows a natural, undetectable hairline, and the transplantation of large numbers of these small grafts during a single session.
Careful patient selection and newer techniques are geared toward naturalness and undetectability for the lifetime of the patient.
In the majority of patients, hair transplantation is performed under local anesthesia. Lidocaine is the most commonly used anesthetic. The key to success is superficial infiltration of the dermis, which allows both better pain control and hemostasis for the patient. The patient is placed in either a prone position or a seated position for donor site removal. The amount of hair in the donor area is the only limiting factor in hair transplant surgery. Therefore, efficiency in harvesting follicular groupings is critical. There are two types of methods for harvesting donor hair: strip harvesting and follicular unit extraction.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree