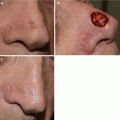
Fig. 5.1
Lentigo maligna. Severe solar elastosis with increased density of junctional melanocytes (a), asymmetric nests (a–c), adnexal involvement (a, b, d), and cytologic atypia of melanocytes (c, d)
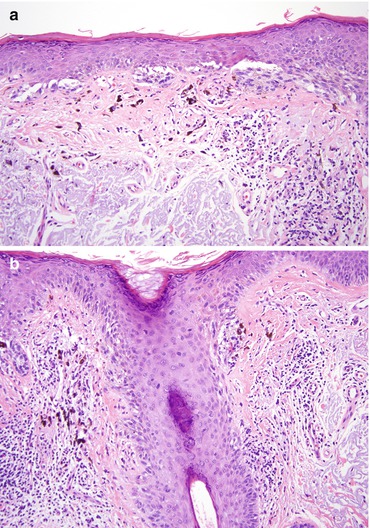
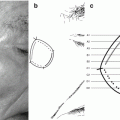
Fig. 5.2
Lentigo maligna. Severe solar elastosis with irregular nests (a, b) also involving the adnexal epithelium (b), and dermal inflammatory infiltrates with melanophages (a, b)
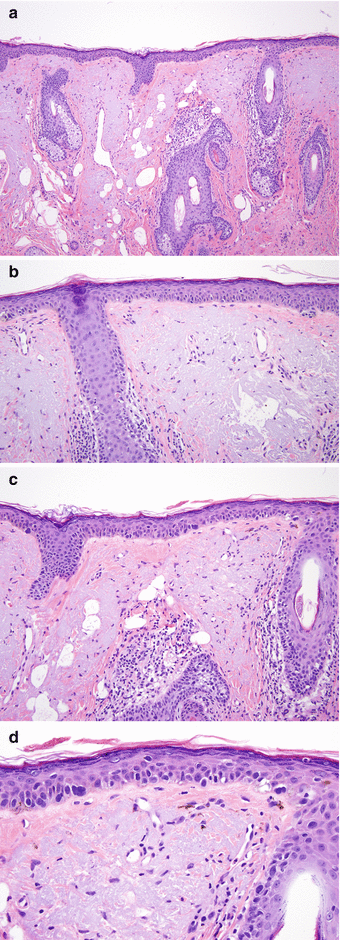
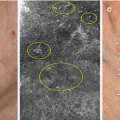
Fig. 5.3
Lentigo maligna. Severe solar elastosis with increase in mostly solitary units of melanocytes in the epidermis and adnexal epithelium (a, c, d), with asymmetric nests of small melanocytes (b), some multinucleated melanocytes (c, d), and pagetoid growth of single melanocytes (c, d)
The first clue to the diagnosis of LM is an increased density of junctional melanocytes [1, 4], typically associated with a solar lentigo-like background, i.e. hyperpigmentation of basilar keratinocytes. The neoplastic melanocytes may be cytologically bland. Not uncommonly, however, there is some nuclear atypia [1] (Figs. 5.1 and 5.3), ranging from slight nuclear enlargement to two or three times the size of a normal melanocyte. Nuclear enlargement is often associated with hyperchromasia [1].
The density of junctional melanocytes varies. While in early lesions solitary units of melanocytes may be separated from each other by a few keratinocytes, in more developed lesions, melanocytes form strips or files and become confluent [1, 4], i.e. form junctional aggregates without interspaced keratinocytes. A conspicuous cytoplasmic fixation retraction artifact producing a clear halo may be present. Neoplastic melanocytes commonly extend into adnexal structures [15] (Figs. 5.1, 5.2, and 5.3). Adnexal involvement is most often first seen in the infundibular portion of the follicle. However, melanocytes may extend deeper to the level of the sebaceous gland and inferior portion of the follicle [4]. This may occasionally cause challenges in assessing dermal invasion and tumor thickness. Junctional tumor growth may lead to the formation of junctional nests. Melanocytes may also be found in the spinous layer [1].
The epidermis of LM is often atrophic with flattening of the rete ridges [12, 17] (Fig. 5.3). The dermis often contains a patchy or band-like lymphocytic infiltrate with melanophages (Fig. 5.2). Some authors suggest that the presence of melanophages is a helpful clue against reactive melanocytic hyperplasia of chronically sun-damaged skin [4]. However, this does not apply when one considers a lichenoid keratosis in the differential diagnosis, since melanophages are commonly seen in this benign condition, which may also be associated with a reactive melanocyte hyperplasia.
In more developed lesions of LM, the nuclear atypia of melanocytes may become more prominent characterized by an angulated or somewhat spindled shaped nucleus [1]. The nuclear chromatin pattern is typically dark (hyperchromatic) [1]. Some of the melanocytes are multinucleated with prominent dendritic processes, a phenomenon, which has been called “starburst giant cell” [18]. The greater the number of nuclei within a multinucleated giant cell, the more likely it is associated with LM [18]. However, the starburst giant cell is not specific for LM, but can also be seen in benign melanocytic nevi, including junctional nevi [19]. Additionally, some melanocytes, possibly already in initial stages of LM, show dendritic appearance, with thick dendrites reaching the upper spinous layer [4, 11].
Associated invasive melanoma may have many different appearances from amelanotic (common) to pigmented (uncommon), epithelioid, fusiform or mixed cell type [20]. A minor degree of stromal fibrosis is often a clue to early superficial invasion. Superficial invasion is often limited to a few isolated tumor cells or small cell aggregates in the papillary dermis. If they are cytologically bland and form nests, they may be difficult to distinguish from a small nevus remnant. Subtle invasive melanoma may at times only be located around follicular structures. Invasive melanoma is readily recognizable, if the tumor cells are pleomorphic, mitotically active and form a mass. Vertical growth phase melanoma is defined as cohesive nests, nodules or plaques larger than those within the epidermis and consisting of atypical tumor cells cytologically different from those in the radial growth phase. In lentigo maligna melanoma, the vertical growth phase often shows spindle cell morphology. Invasive tumors associated with LM may or may not display stromal desmoplasia and perineural invasion [20]. If desmoplasia is prominent, the tumor is designated “desmoplastic”. Up to two thirds of desmoplastic melanomas are associated with LM [21–23]. Neurotropism is rare in small superficial tumors, but not uncommon in more deeply invasive LMM [4]. While it is most often seen in association with fusiform cells, epithelioid melanoma may also be neurotropic.
Although the epidermis is typically atrophic, LM may also contain areas with elongated rather than attenuated rete ridges. This may be in some cases due to LM coinciding with a pigmented actinic keratosis, solar lentigo, or seborrheic keratosis [24]. Some authors describe these lesions as simulating a dysplastic nevus or a lentiginous nevus. Based on one study, dysplastic nevus–like areas, defined as having elongated and/or fused rete ridges and prominent medium to large nests of pleomorphic melanocytes with little or no pagetoid upward migration, predominated in as much as 43 % of LM/LMM [25]. Some authors have suggested a term lentiginous melanoma for lesions with features of LM but with preservation of rete ridges and absence of prominent solar elastosis [26]. It is important to recognize these variants, especially if examining a partial biopsy to avoid a pitfall of erroneous diagnosis of a dysplastic nevus or a lentiginous nevus.
Ancillary Diagnostic Test
Immunohistochemistry
Immunohistochemical stains for melanocyte antigens may facilitate a more accurate assessment of melanocyte density and growth pattern. This is specifically useful if a dense inflammatory infiltrate is present, pseudonests are suspected, or if a distinction between LM and reactive melanocyte hyperplasia due to chronic sun damage or prior surgery is difficult [27]. A pseudonest or a pseudomelanocytic nest is an aggregate of cells and cell fragments, including keratinocytes and inflammatory cells, and occasional melanocytes, that may mimic a melanocytic proliferation. The histologic evaluation of surgical margins, where the utility of immunohistochemical markers may be particularly useful in certain instances, is covered elsewhere in the textbook.
Melanocyte antigens include S100 (anti-S100 antibody), SOX10 (anti-SOX10 antibody), gp100 (HMB-45 antibody), Melan-A or MART1 (anti-Melan-A, anti-MART1, or A103 antibodies), microphthlamia transcription factor (MITF) (anti-MITF antibody), tyrosinase (anti-tyrosinase), and nerve growth factor receptor (NGFR) (anti-NGFR antibody) [28]. Of these, S100 protein, SOX10, and NGFR are particularly helpful for the detection of desmoplastic melanoma [29], but they are the least “specific” markers, because the respective antigen is also expressed by a number of non-melanocytic cells [28]. Caution is necessary when calibrating immunohistochemical stains. Tyrosinase and Melan-A/MART1 are sensitive markers for melanocytes, however, in many laboratories the suboptimal use of immunohistochemical markers with cytoplasmic stains may overestimate the density of melanocytes [27, 30, 31]. The use of nuclear markers (MITF or SOX10) avoids that pitfall, but comes at the expense of potential “false-positive” interpretations of non-melanocytic dermal cells, which may be positive for MITF and SOX10. Due to limited sensitivity the use of HMB-45 may result in underestimation of intraepidermal melanocyte density [32, 33].
At present, there are no markers to discriminate between benign and malignant melanocytes. Recently, some have suggested that the differential localization of soluble adenylate cyclase (sAC) in LM versus benign melanocytes may be a useful diagnostic adjunct for the diagnosis of LM [34]. However, we have not found this marker to be reliable or useful (KJB, 2015, personal observations). Normal sun-damaged melanocytes may show nuclear expression of sAC. In our experience the vast majority of cases of lentigo maligna can and should reliably be diagnosed by examining one or a few H&E-stained sections. The routine use of immunohistochemical markers is unnecessary, and may at times lead to overdiagnosis.
Immunohistochemical markers, however, may help identify a dermal invasive component, such as a subtle desmoplastic melanoma, and help distinguish invasive melanoma from a histologic simulant, such as in the scenario of LM colliding with an atypical fibroxanthoma. Immunohistochemical stains may also on occasion help distinguish a nevus remnant from a nevoid invasive melanoma. If, for example, a nevoid dermal population is immunoreactive for BRAFV600E while the associated lentigo maligna melanoma in situ is BRAF-negative, one can reasonably conclude that the nevoid population is unrelated to the lentigo maligna and unlikely to represent an invasive LMM. While BRAF V600E mutations would be very unlikely in a LMM [35, 36], some tumors carry an NRAS mutation. An immunohistochemical stain for NRASQ61R can help document the presence of such a mutation for treatment purposes.
Molecular Tests
The diagnosis of LM/LMM is usually made by histopathologic examination. Molecular tests may be considered for rare invasive tumors with microscopically ambiguous or controversial features and/or to determine eligibility for targeted therapies for metastatic melanoma. Currently, the molecular tests used for diagnosis include cytogenetic methods, such as fluorescence in situ hybridization (FISH) and comparative genomic hybridization (CGH) [37] and possibly gene expression profiling [38, 39]. Mutation analysis is indicated to guide targeted therapy of metastatic melanoma, especially to determine the presence or absence of BRAF (B-Raf Proto-Oncogene, Serine/Threonine Kinase) V600 codon mutations [40, 41]. Prognostic gene expression panels have also been proposed, but are associated with methodological shortcomings and have not yet been validated [38, 39, 42].
Differential Diagnosis
Melanocytic Hyperplasia of Sun-Damaged Skin
The number and size of melanocytes can be slightly increased in benign chronically sun-damaged skin [43]. The melanocytes should, however, be regularly distributed along the basal layer at an equal distance from one another [4] (Fig. 5.4). Extension to the infundibulum of the hair follicle may be seen, but the increase in melanocytes should not involve the inferior part of the hair follicle. Nests are not present and starburst giant cells are rare.
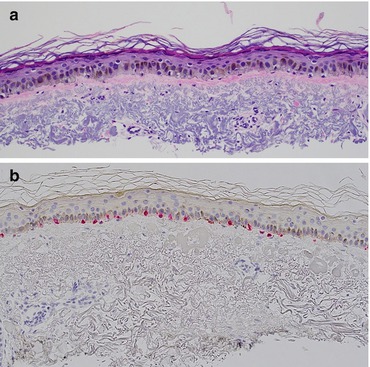

Fig. 5.4
Solar lentigo with melanocytic hyperplasia. (a) Basal layer hyperpigmentation with a slight increase in the density of cytologically bland melanocytes. (b) An immunostain for melan-A documents a slight increase in the density of junctional melanocytes
The average number of melanocytes of non-lesional skin from the head/neck skin is approximately 9–10 melanocytes per 0.5 mm on H&E (hematoxylin & eosin)—stained sections and 12–15 per 0.5 mm using Melan-A/MART1 immunohistochemistry [44–47]. Continuous melanocytes, atypical melanocytes, and follicular extension can occasionally be seen in the sun-damaged skin surrounding melanoma or non-melanoma skin cancer [46]. In one study, one third of the non-lesional skin specimens showed areas with moderate confluence with 3–6 adjacent melanocytes based on MART1 immunohistochemical staining [44]. Melan-A/MART1 may lead to overestimation of the density of melanocytes, especially when the immunohistochemical methods are suboptimal [33]. Nuclear staining (MITF, SOX10) allows a better distinction between a keratinocyte and a melanocyte and may therefore be the preferred method for measuring melanocyte density within the epidermis. The most helpful features suggestive of LM other than an increased number of solitary units of melanocytes include nests of melanocytes, especially unevenly placed nests, irregular distribution of melanocytes, including deep extension of melanocytes along the adnexal epithelial structures, irregular distribution of melanin pigment, presence of melanocytes above the junction, and atypical nuclei [47].
Solar Lentigo
Solar lentigo may be difficult to distinguish from early LM, especially if there is a background of solar melanocyte hyperplasia (melanocytic hyperplasia of chronically sun-damaged skin). In general, solar lentigo shows normal to only slightly increased melanocyte density. The mean melanocyte count in solar lentigines is significantly lower than in fully evolved melanoma in situ, 27 versus 112 per 1 mm, respectively [33]. Additionally, confluence of melanocytes, nests of melanocytes, growth along the deeper portions of adnexal structures, and pagetoid melanocytes are absent in solar lentigines [48] (Fig. 5.5a). Melanocyte atypia can be seen if superimposed melanocytic hyperplasia of chronically sun-damaged skin is present. Rete ridges are typically normal or elongated, although focal effacement may be present. Knowledge of the clinical size and complexity of the lesion is imperative for correct pathologic diagnosis.
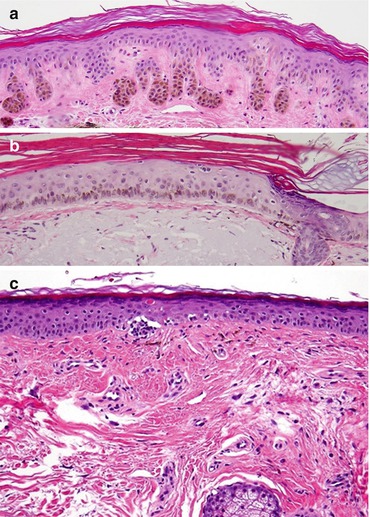

Fig. 5.5




Differential diagnosis of Lentigo maligna: (a) Solar lentigo, (b) Pigmented actinic keratosis, (c) Lichenoid Keratosis
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree