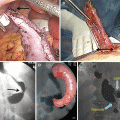
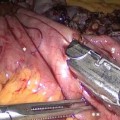
Fig. 19.1
Position and port placement
Rather than use external landmarks such as the umbilicus or xyphoid to determine port placement, we feel it better to place the trocars based on internal anatomy and visceral relationships. In this way, triangulation and visualization will be preserved, accommodating for variations in the size of the liver, or length of the patient’s torso.
Attention must not only be given to individual trocar placement, but also the angle in which the trocar enters the skin. Some individuals’ thick, muscular abdominal walls do not allow for the range of motion necessary to achieve the objective, forcing redirection of the trocar internally, through the same skin incision, but different fascial opening, or by placement of another trocar. In general, the optimal placement is to orient all trocars toward the midline, pointing to the base of the mesocolon.
Extra long trocars may be necessary and although some surgeons prefer to limit the number of 12 mm trocars (necessary to accommodate stapling devices), this may limit proper stapler orientation and compromise the anatomic construct. The hernia risk is minimized by either closing the trocar defects, or preferably, using non-bladed trocars without fascial closure to minimize postoperative pain.
Our port placement scheme is as follows; it illustrates the rationale necessary for consistency of this technique and represents an evolutionary process that has taken over 12 years to develop (Fig. 19.1).
Initial trocar (12 mm) – Left, upper quadrant, subcostal, mid-clavicular line. This is often an optical entry without prior insufflation. The rationale is that many patients have had previous procedures, pelvic or otherwise—this area is rarely affected with intra-abdominal adhesions from common open procedures. This allows dissection of midline adhesions, inspection of the size of the liver, and determination of the best level for the primary optical port. This will also be the primary port for vertical stapling of the gastric pouch. Once adhesions are mobilized, then the optical port can be thoughtfully placed as to see the ligament of Treitz as well as hiatus without having to “turn around.” Also, by keeping the initial entry away from the midline, the vena cava and aorta are not as vulnerable to injury.
Primary optical trocar (12 mm) – Placement has been described above. Optimal placement allows for forward visualization of the proximal small bowel and the hiatus. Once this trocar is placed, the camera is moved to this port for subsequent trocar placement. I have not found the current 5-mm scopes to provide enough light delivery and therefore resolution for optimal visualization in most patients.
Right–sided trocar (12 mm) – This trocar must be placed thoughtfully just as all others. Exterior landmarks are irrelevant. It must come in below the liver edge, just to the right of the midline so as to be able to triangulate on the hiatus as well as the ligament of Treitz; therefore, it should be angled toward the root of the mesocolon, rather than perpendicular to the abdominal wall. It must be 12 mm to accommodate the stapler that will define the inferior gastric pouch.
Left–inferior trocar (12 mm) – This is often at the same level as the primary optical trocar and in the same line as the initial trocar. This will be the primary stapler entry site for the jejuno-jejunostomy and along with the right-upper quadrant trocar will triangulate very well for a comfortable manual gastro-jejunostomy.
Liver retractor – The most optimal placement appears to be subxyphoid. A 5-mm port can be used here, depending on the liver retractor of choice. We have found that a simple 5-mm instrument or similar device will provide excellent exposure and therefore is often placed without a trocar, through direct puncture, as it will not be removed until the end of the case.
The omentum is displaced cephalad to expose the ligament of Treitz. In patients whose omentum is adherent to pelvic structures or involved in an incarcerated ventral hernia, we prefer to incise the gastrocolic omentum and open the transverse mesocolon from above, thus exposing the ligament of Treitz directly. Ventral hernias are repaired at a later date when optimal weight loss and nutrition ensure a greater degree of primary success and the use of prosthetic mesh is not compromised by contamination of enteric contents.
The proximal jejunum is transected with a 2.5- to 3.8-mm linear stapler, depending on the thickness of the bowel, and the mesentery is divided with another firing of the stapler or with an ultrasonic scalpel. The Roux limb is measured and a side-to-side linear anastomosis is performed (Fig. 19.2). Typically, the length of the Roux limb can be up to 150 cm without an associated increased incidence of malabsorptive complications [24]. The enterotomy is closed with a single layer of absorbable suture. The mesenteric defect must be closed with a continuous, nonabsorbable suture to limit the possibility of internal herniation (Fig. 19.3).
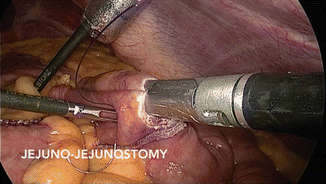
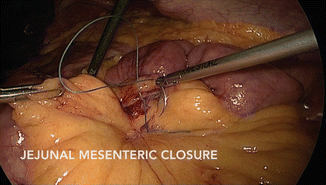

Fig. 19.2
Jejuno-jejunostomy

Fig. 19.3
Jejunal mesenteric closure
The Roux limb is passed through a retrocolic tunnel and fixed to the transverse mesocolon with nonabsorbable sutures, which also includes closing the Petersen’s space (Figs. 19.4 and 19.5), again, to help prevent possible internal herniation. Alternatively, some surgeons prefer an antecolic route for the Roux limb, claiming a lower incidence of postoperative bowel obstructions [25].
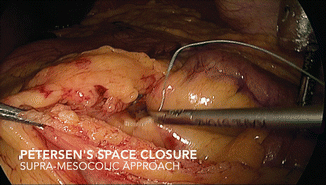
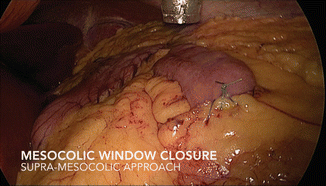

Fig. 19.4
Petersen’s space closure

Fig. 19.5
Mesocolic window closure
There are times when the mesocolon is uncomfortably short and will not allow for the safe passage of a retrocolic Roux limb. In these rare instances, the decision to route the Roux limb antecolic must be made before the transection of the jejunum. This site must be more distal from the ligament of Treitz, typically 50–100 cm, to limit the tension on the gastro-jejunal anastomosis. By lengthening the biliopancreatic limb, iron and calcium absorption may be less efficient, and the incidence of these deficiencies may be theoretically increased or more difficult to manage with oral supplementation alone.
Controversy exists as to whether the large resultant Petersen’s space associated with an antecolic Roux limb requires closure. Clearly, these patients are still at risk for intestinal volvulous [26]. Therefore, our philosophy is to eliminate the risk of postoperative bowel obstruction rather than simply settling for a reduction in the incidence. However, the long-term stability of suture closure of these defects is still to be determined.
The liver retractor is now placed to allow dissection of the proximal stomach. Occasionally, a very large liver will not allow for sufficient visualization—an indication for open conversion. However, displacement of the liver to the right, rather than anterior, will allow sufficient exposure in the largest of patients. Alternatively, the surgeon may decide to abort the procedure, evaluate the cause of hepatic enlargement (usually steatosis), and institute therapy (medical weight reduction) in anticipation of performing the procedure at a later time under more ideal circumstances. In this way, surgical restraint and proper judgment may reduce the morbidity associated with these operations.
The pouch is formed by sequential firing of a laparoscopic linear cutter, stapling device around a 34 Fr. orogastrically placed bougie. The first firing is horizontal, introducing the stapler from the RUQ port (Fig. 19.6), beginning no more than 5 cm distal to the esophagogastric junction, using 3.8- to 4.1-mm cartridges, depending on stomach thickness. It is important to orient the stapler slightly cephalad to angulate the stapler line. Subsequent firings are vertically oriented to the angle of His, introducing the stapler from the LUQ port (Figs. 19.7 and 19.8). By using this port configuration, extra-long staplers are unnecessary for the majority of patients.
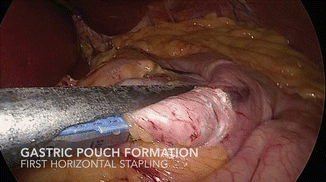
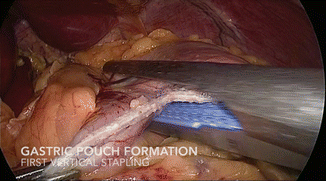
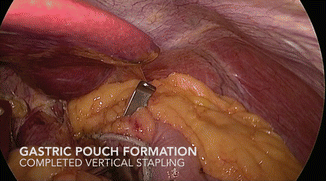

Fig. 19.6
Gastric pouch formation

Fig. 19.7
Gastric pouch formation

Fig. 19.8
Gastric pouch formation
One of the critical steps in creation of the gastric pouch is posterior visualization at the level of the hiatus. Many surgeons will “bluntly” dissect behind the stomach, but given the variable level of adhesions to the pancreas and the splenic vessels, this is unwise. Optimally, it is better to enter the lesser sack through the gastrocolic omentum and free the posterior gastric adhesions up to the esophageal hiatus. This protects the pancreas and the occasional tortuous splenic artery from inadvertent injury. After this, the lesser curve, perigastric dissection can be performed with more confidence and placement of the stapler more precisely as not to “twist” the stomach pouch. This occurs when posterior gastric adhesions prevent the initial horizontal stapler from capturing equal amounts of anterior and posterior gastric wall. The resultant twist is not as critical as in the gastric sleeve but is not esthetically pleasing.
Controversy exists as to whether or not to dissect the hiatus and repair a hiatal hernia when present. Autopsy studies show that a hiatal hernia is present in up to 70 % of individuals, similar to our observations. However, dissection of the hiatus can add additional time and potential complications to an already complicated procedure. Our studies have not shown that preoperative endoscopy accurately predicts the absence of a hiatal hernia; the only way to determine its presence is circumferential dissection of the esophagus. The absence of the “anterior” dimple is not reliable as the hernia space is often taken up by a large para-esophageal lipoma that can be easily reduced into the abdomen once identified. Once identified, the hiatal hernia is best repaired posteriorly (Fig. 19.9).
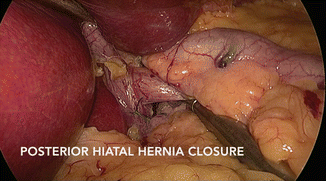

Fig. 19.9
Posterior hiatal hernia closure
The question remains: “Is it important to repair every hiatal hernia at the time of gastric bypass?” The answer is not clear. If one assumes that precise dissection and formation of the gastric pouch is important to limit postoperative complications, then it would be appropriate to absolutely identify the location of the gastroesophageal junction, often hidden in a “sea of fat,” to better perform more consistent reconstruction. It has been our observation that almost 100 % of patients we have reoperated after gastric bypass have a significant hiatal hernia at the time of reoperation – something not appreciated at the time of the first intervention.
Dissection of the hiatus and repair of the hiatal hernia along with removing the fat pad overlying the angle of His may allow for more precise and consistent pouch formation and subsequent better long-term performance and lower complications, but this has not been proven. Although this may add up to 5–10 min of operative time, which is significant, the added exposure may make for a safer operation. Each surgeon will need to evaluate this perception in the context of his or her individual experience and skill level. Suffice it to say, to perform a good laparoscopic gastric bypass, the surgeon must be expert at hiatal dissection.
Routing of the Roux limb is best determined by the patient’s internal anatomy. As stated previously, the risk of internal herniation is the same, whether routed antecolic or retrocolic as long as potential internal hernia spaces are closed with nonabsorbable sutures. It is important that there is as little tension on the anastomosis as possible. In most cases, an antegastric orientation makes gastro-jejunal anastomosis more visible and easier for subsequent revision if necessary. However, the retrogastric orientation is a more direct route and offers unobstructed access to the anterior stomach.
The method of gastro-jejunal anastomosis is not critical. Whether stapled, or hand-sewn, primary or reinforcing, sutures should be absorbable to prevent suture migration or ulcer formation. Choice of suture material, braided or monofilament, single- or two-layer, is left to the discretion and results of each individual surgeon (Figs. 19.10, 19.11, 19.12, 19.13, 19.14, and 19.15).
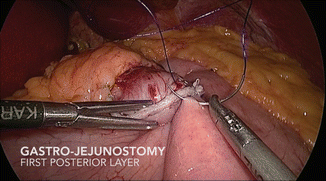
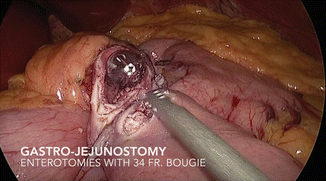
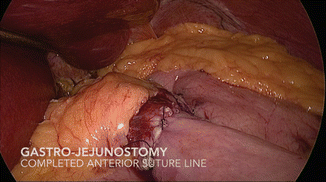
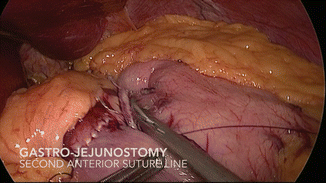
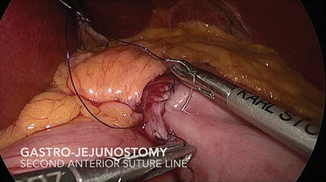
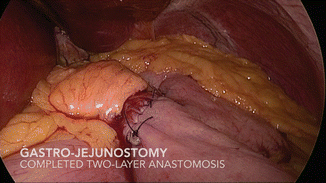

Fig. 19.10
Gastro-jejunostomy

Fig. 19.11
Gastro-jejunostomy

Fig. 19.12
Gastro-jejunostomy

Fig. 19.13
Gastro-jejunostomy

Fig. 19.14
Gastro-jejunostomy

Fig. 19.15
Gastro-jejunostomy
The anastomosis and proximal staple lines can be tested with blue dye, air insufflation via the orogastric tube, or operative endoscopy. Routine drainage is unnecessary. The port sites are inspected for bleeding on withdrawal of the trocars and the skin is closed with simple absorbable monofilament sutures.
19.3 Postoperative Management
Perioperative antibiotic is continued for 24 h, while thromboembolism prophylaxis continues until the patient is discharged. Analgesia is in the form of patient controlled narcotic delivery systems and intravenous ketorolac or acetaminophen. Oral narcotics are offered when clear liquids are tolerated. Metoclopramide is administered routinely and a variety of antiemetic pharmacologic agents are available for nurses to use at their discretion.
Routine postoperative contrast studies add little to the management of these patients and serve only to delay discharge secondary to nausea [27]. A normal postoperative upper gastrointestinal (UGI) study should not preclude the surgeon’s intervening based on clinical suspicion of a leak [28].
Patients are started on clear liquids the day of surgery and are required to ambulate with assistance. Preoperative oral medications can be resumed as soon as the patient can tolerate clear liquids. Most patients are discharged by the second postoperative day, the majority on the first postoperative day.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree