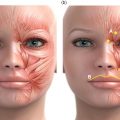
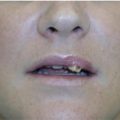
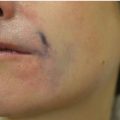
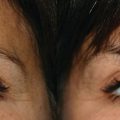
4 Tirbod Fattahi and Salam Salman Department of Oral and Maxillofacial Surgery, University of Florida, Jacksonville, FL, USA History of hyaluronic acid (HA) dermal fillers goes back to the late 1970s when the science of injectable skin fillers really began. Although the first HA filler was not formally introduced in the USA until the early 2000s, bovine collagen became the first injectable filler material in 1977 when Zyderm and Zyplast (Inamed Aesthetics, Santa Barbara, California) were introduced. Both derived from cow collagen, Zyderm and Zyplast became quite popular simply due to absence of any other competitor and their effectiveness in eliminating moderate to deep rhytids and augmenting soft tissue defects. Similar to today’s HA fillers where scientific modification (crosslinking) of HA particles produces different products within the same family (Juvéderm Ultra vs. Juvéderm Ultra Plus), Zyplast was formulated to be the more “viscous” formulation, indicated for a deeper level of augmentation. The biggest drawback of bovine collagen injectables became the need for allergy testing. Although further enhancements of bovine collagen were performed (Zyplast 2), bovine collagen injectables eventually lost their luster and are no longer on the market. However, many manufacturers, both national and international, continued to look for safer and newer options for injectable dermal fillers. Finally, in 2003, Restylane (Galderma Laboratories, Fort Worth, Texas) became the first injectable HA filler approved in the USA. Since then, there have been over a dozen other HA dermal fillers that have entered the cosmetic surgery market with various degrees of success. The term HA is derived from hyalos (Greek for vitreous) and uronic acid since it is abundantly found in the vitreous fluid of the eye with a high concentration of uronic acid. HA, is a naturally occurring protein (glycosaminoglycan) component of the extracellular matrix throughout the human body. The HA peptide is a combination of repeating sequences of N‐acetyl‐glucosamine and glucuronic acid. The highest concentration (over 50%) is found in human skin, eyes, as well as joint spaces. It is estimated that a 70‐kg male adult has about 15 g of HA in his body [1]. Because it is a hydrophilic protein, HA attracts water and maintains hydration wherever it is injected or naturally resides; this characteristic has high clinical value which will be discussed later. All HA particles also have rheologic properties (elasticity and viscosity) which is the critical advantage in management of osteoarthritis, one of the earliest indication for HA injectable therapy. The main type of HA used in cosmetic surgery today is known as Non‐Animal Stabilized Hyaluronic Acid (NASHA). This formulation, as the name implies, does not have any animal products unlike some earlier HA injectables used in the treatment of retinal issues or arthritic conditions. This type of HA filler is typically formulated in a gel‐like consistency. The viscosity of this gel‐like substance is directly influenced by cross‐linking, the process of attaching multiple chains of HA together. Cross‐linking will increase the particle size of HA fillers, which will increase their longevity and will make the fillers more viscous. More cross‐linked fillers are more appropriate for deeper plane injections. The most commonly used HA fillers in the USA are the following: Many of the manufacturers have created multiple types of fillers within the same family by modifying the cross‐linking and/or particle size; this modification allows for various types of clinical indications. For example, Juvéderm fillers are formulated as Ultra, Ultra Plus, or Voluma, all depending on the degree of cross‐linking and particle size. Similarly, Restylane has been formulated as Restylane, Restylane Silk, and Restylane Lyft, again depending on particle size, cross‐linking, and clinical indications. Local anesthetic has also been added to the formulation of many HA fillers for pain control during the injection process. For example, Juvéderm Ultra XC contains xylocaine®. HA dermal fillers are indicated for volume replacement and/or augmentation at the recipient site. As with most cosmetic injectables, including neurotoxins, most indications are not Food and Drug Administration (FDA) approved and are considered off‐label use. It is up to the imagination of the clinician, the characteristics of the specific filler, and the location of the defect that determine the specific type of HA to use. Most common indications for HA fillers in the face include: Dermal fillers, including HA fillers, as implied in their name, are indicated for dermal and/or deeper plane injection. There are several different techniques of depositing the filler. Serial injections of small depots of filler is probably the most commonly used technique, especially along linear regions (lip liners, nasolabial grooves). Cross‐hatching or injections along 2 vectors at 90′ to each other works well in larger, flat areas (cheeks, tear troughs). This allows “stacking” the filler in multiple planes for a better outcome. Regardless of the technique, injections are in the dermal or deeper plane with deposition of the HA filler in a retrograde fashion as the needle is being withdrawn. If the injection is too superficial, simply massage the filler out of the area. The phenomenon of “Tydnall effect” can occur when any particle absorbs ambient light and reflects a “bluish” color (Figure 4.1). This is applicable to an excessively superficial (epidermis) injection of HA fillers; particles of fillers will absorb light and display a blue discoloration. Patients often think this is a bruise; however, it does not resolve until the filler particles have completely dissolved. Management of the Tyndall effect will be discussed later in the chapter. Figure 4.1 Tyndall effect; bluish hue as light is absorbed by particles.
Hyaluronic Acid Dermal Fillers
4.1 Introduction
4.2 Hyaluronic Acid
4.3 Available Products
4.4 Clinical Indications
4.5 Injection Techniques

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree