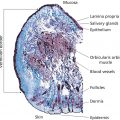
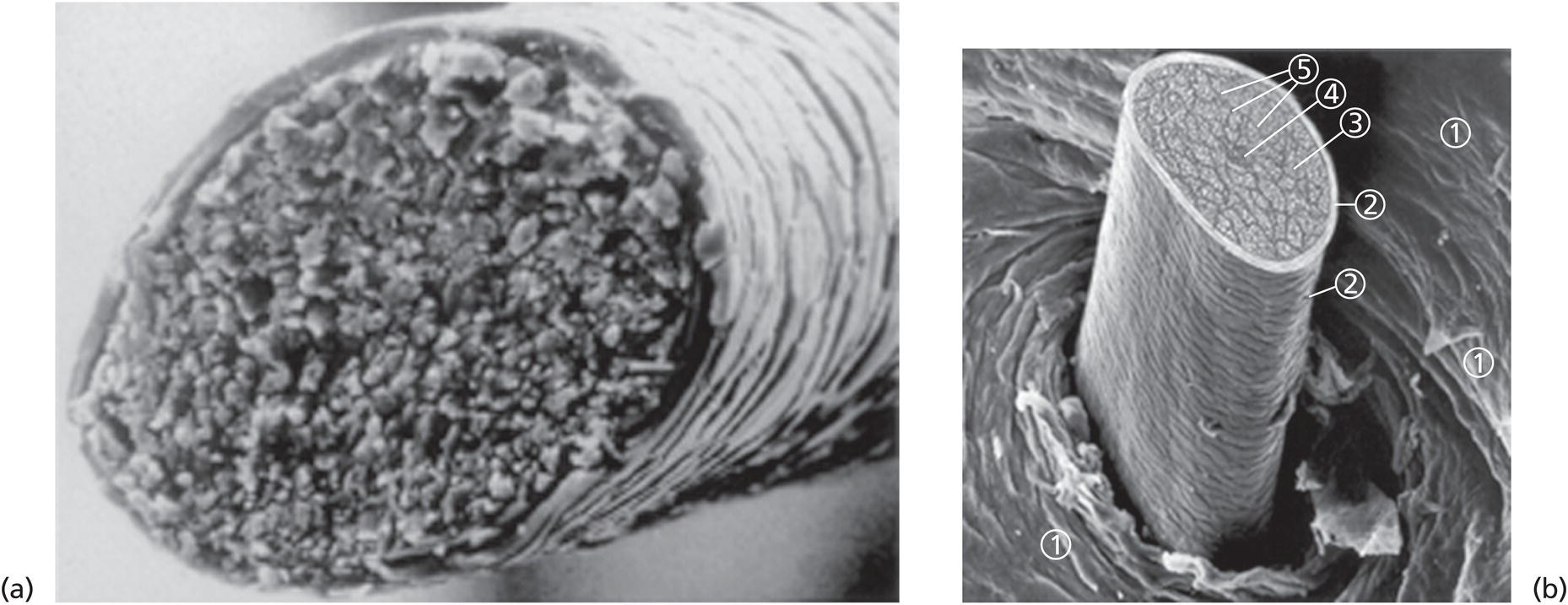
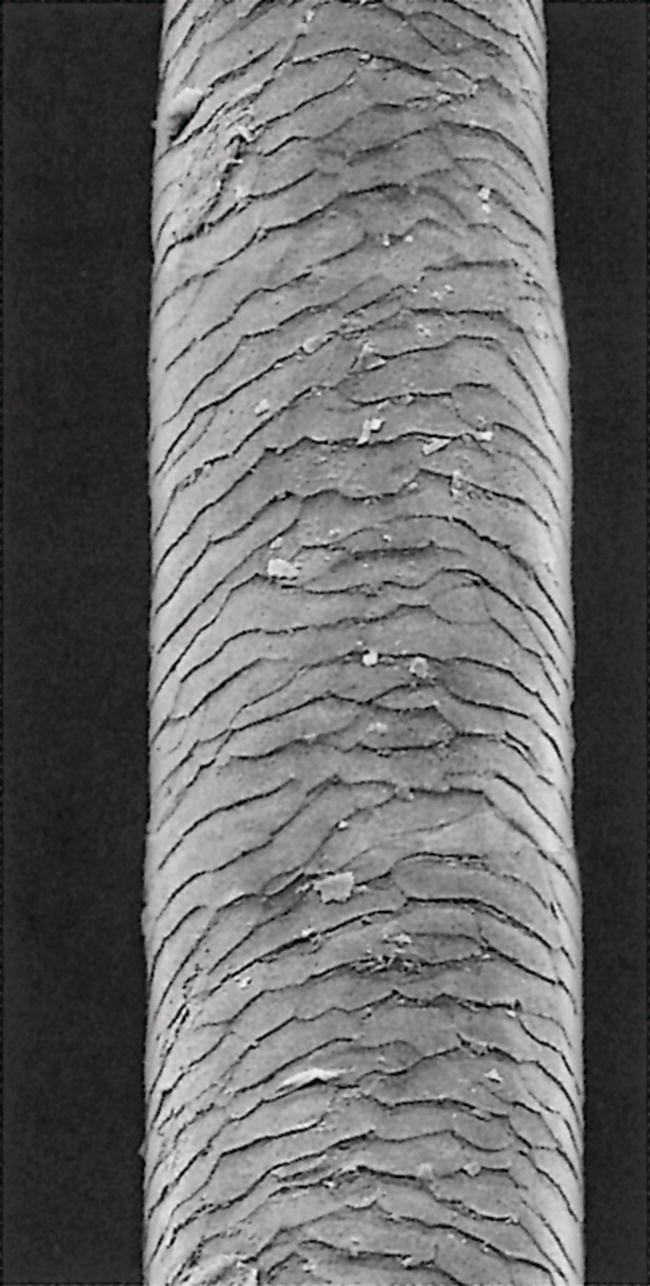
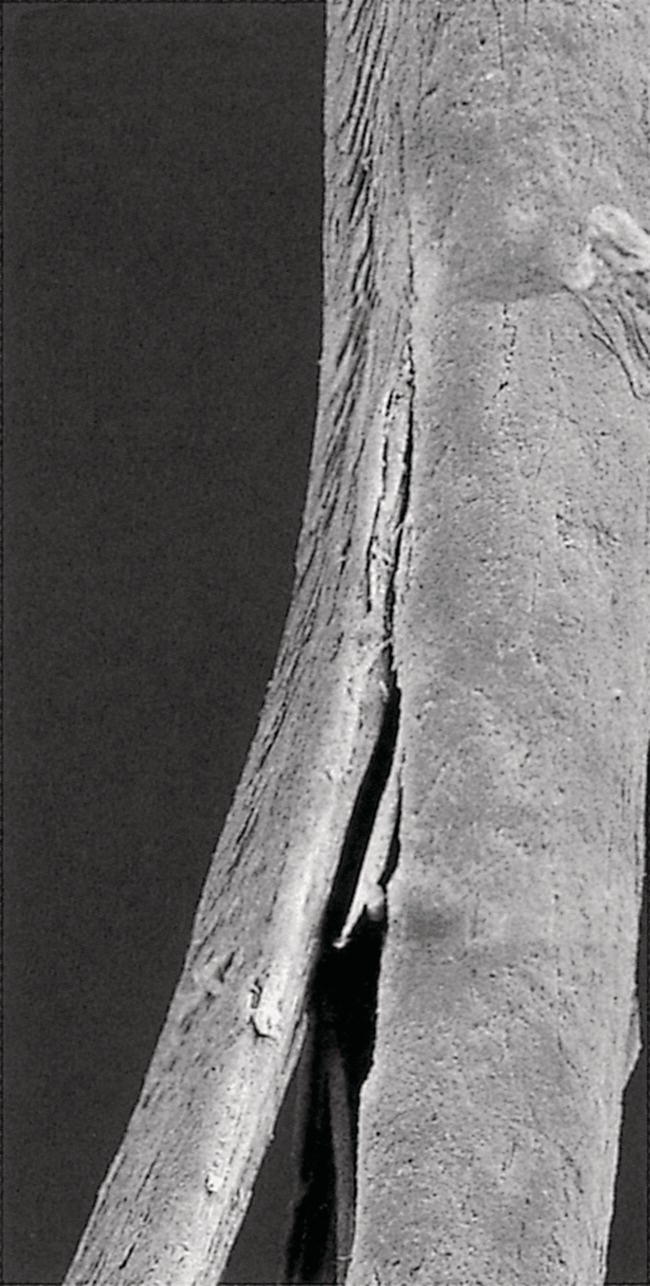
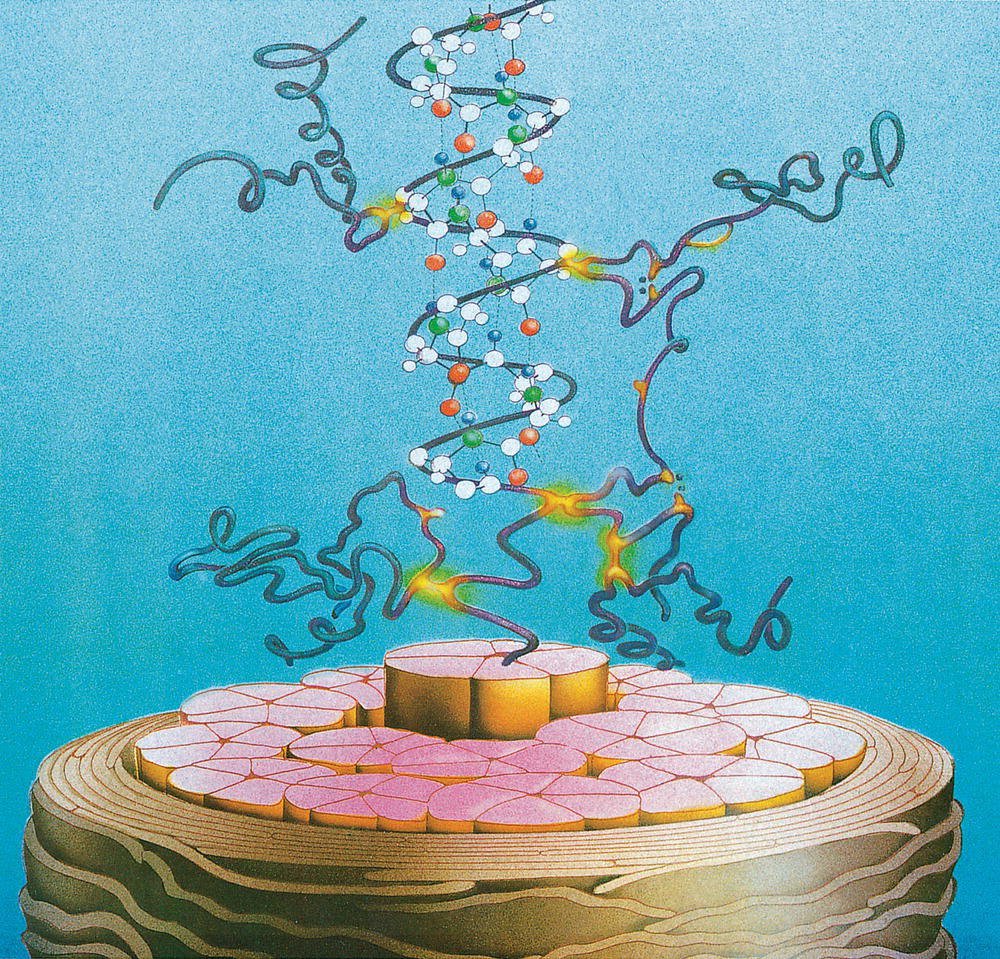
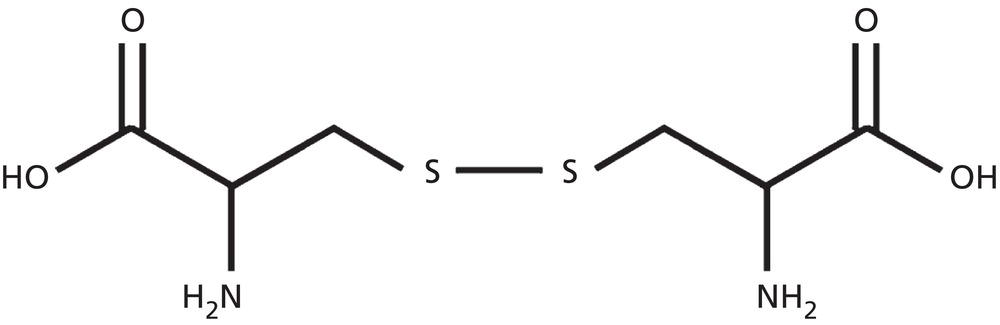
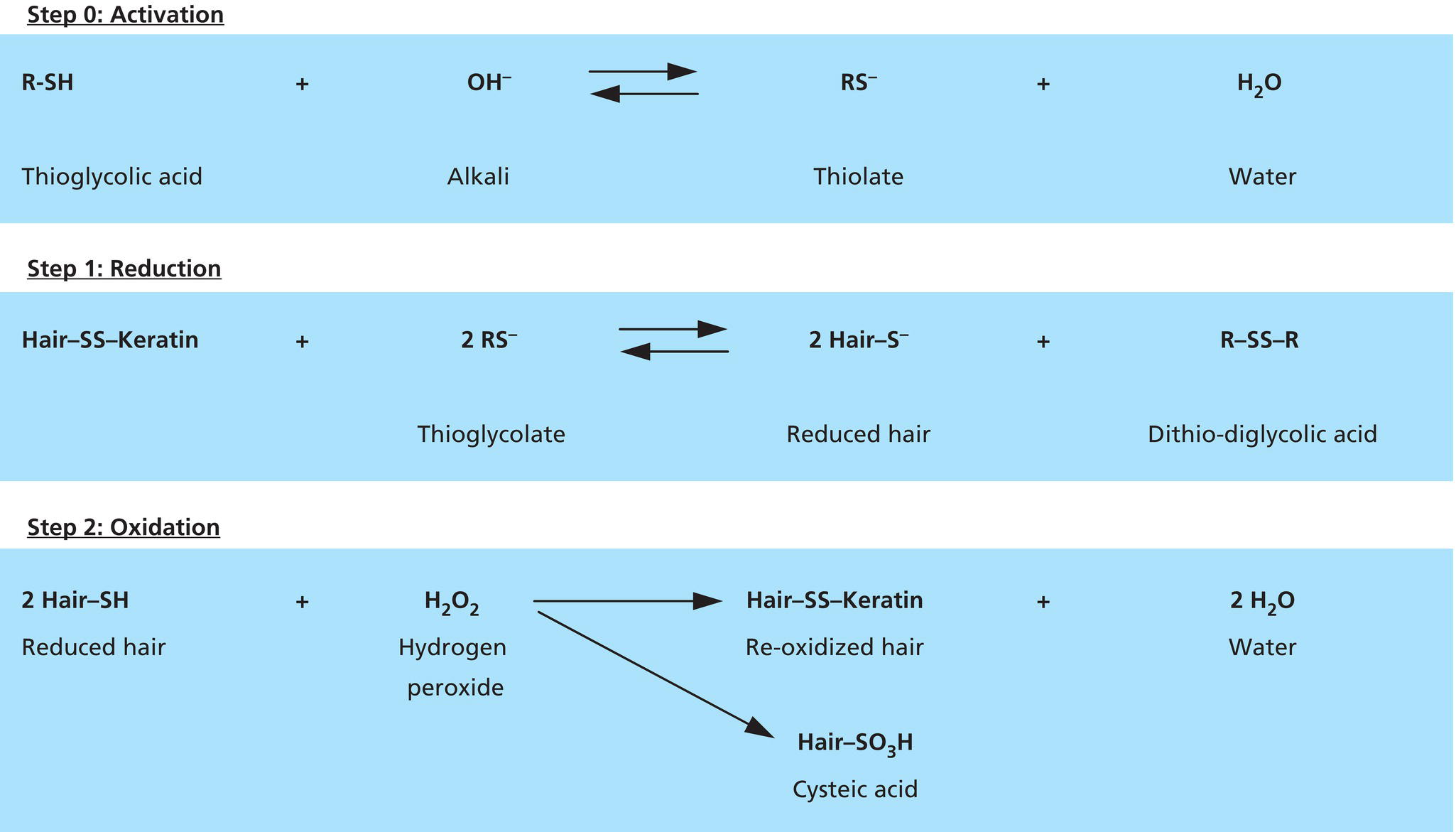
Annette Schwan‐Jonczyk1, Gerhard Sendelbach2, Andreas Flohr3, and Rene C. Rust4 1 Private Practice, Darmstadt, Germany 2 Darmstadt, Germany 3 Wella/Procter & Gamble Service GmbH, Darmstadt, Germany 4 GSK/Stiefel, Brentford, Middlesex, UK Since ancient cultures, curly hair has been regarded one of the most prominent signs of femininity and beauty. Women with straight hair purchased expensive wigs or spent hours for hair ondulation with water and heat for a temporary success. A ground‐breaking invention was made by Carl Nessler in 1906 who offered, for the first time, irreversible hair shaping to clients by means of heat and borax. Improvements followed during the twenthieth century by the creation of “cold waves,” using sulfite or thioglycolate as actives, still the most popular waving agents in home and salon perms. Hairs are composed of cells packed in tight cell bundles that grow out from up to 3 mm skin depth. About 5 million hairs cover the human body and scalp classified as vellus hairs (5–10 μm in diameter) or terminal hairs (5–120 μm in diameter). The human head has 100,000–50,000 fibers, which grow approximately 1 cm per month through rapid cell division in the living, lowest part of their hair follicle, known as the hair bulb. Each strand of hair is made up of three regions, the medulla, the cortex, and cuticle. The innermost structure of the hair, the medulla, is not always present and is an open, spongiform region. The highly structural and organized cortex, or middle layer of the hair, is the primary source of mechanical strength and water uptake. The cortex contains melanin, which colors the fiber based on the number, distribution, and types of melanin granules. It is currently under discussion if either the shape of the follicle determines the shape of the hair, or if the way the hair forms and hardens determines the shape of the hair follicle. There are ethnical differences in hair shape and structure, for example Asian hair typically has a round fiber diameter and is normally straight. Hair fibers with oval and irregularly shaped diameters are generally wavier or even curly. The hair surface contains flattened cells, known as the cuticle, which forms an imbricate structure resembling shingles on a roof. The hair surface is covered with a single molecular layer of lipid (“f‐layer”) which gives the hair surface hydrophobic properties. However, when the hair fiber swells, for example induced by chemical treatments, its complex structure changes and the cuticle layer is opened making the hair more porous. The cuticle provides protection and support for the inner spindle‐shaped “cortical cells,” which make up 80% of the hair mass. During hair growth, the interdigitated cells are organized into a three‐dimensional network resembling a jigsaw puzzle. This construction contributes to cell cohesion and hair fiber strength (Figure 33.1). Figure 33.1 (a) The fracture plane of a hair fiber clearly shows the composite of a fibrillar core with flattened cuticle cell coating (SEM × 1420). (b) Just emerging hair: 1, scalp surface; 2, cuticle pattern of hair fiber surface; 3, interior of the hair with cortical cells; 4, medullary cells; 5, cell membrane. Figure 33.2 The intact cuticle pattern of the hair fiber near the scalp (SEM × 800). While all hair fibers contain a cuticle, only thicker hair fibers with a diameter greater than 75 μm contain a central area of hollow cells, the medulla (Figure 33.2). For animals, the medulla also enhances thermal insulation. The active growth period of scalp hair is known as the anagen phase and is limited to 3–6 years. Then the hair disconnects from the papilla, a period known as the catagen phase, and enters a 2‐month resting phase, known as the telogen phase, and is subsequently lost through shedding. Hair shedding and renewal results in an average loss of up to 100–125 scalp hairs per day. With constant hair growth and no cutting, bulk hair may reach up to 1 m in length – depending on the genetically determined length of the hair growth cycle (for more hair growth details, the reader is referred to more detailed references [1,2]). Usually, scalp hair is visualized as a circular fiber but cross‐sections of individual hairs reveal variation in hair shape. The typical appearance of a person’s hair is determined by its special mixture of thick, thin, elliptical, kidney‐shaped, and triangular cross‐sectional shapes [3]. “Unmanageable” hair often comprises a high percentage of irregular hair shapes. Irregular hair geometry, small diameter, and large hair diameter all create permanent waving challenges. Hair thickness has a large influence on the shape and hold of permanent wave curls as well [4]. Although hair is not soluble in water, its interaction with water is of particular importance for permanent waving [2, 5]. The hair surface is naturally hydrophobic, and permanent waving lotions contain surfactants for enhanced wetting, the first step in the curling process. Under certain climatic conditions, hair adsorbs atmospheric water up to 30% of its own weight. Although feeling dry at ambient temperature, hair still contains 15% water. Excess water can be bound by capillary forces. The hair water content dictates its chemical reactivity as water widens the hydrogen‐bond network of protein side chains and acts as the vehicle for all permanent waving ingredients. Proper water balance is important for a successful permanent waving. Moreover, as water functions as a plasticizer for hair, permed hair loses curls more rapidly after washing or exposing it to humid conditions, which can be measured in a curl retention test. In presence of water, hair displays its amphoteric character. A hair is at the “isoionic point” when the positive and negative charges of hair proteins are at an equilibrium. The natural “intrinsic point of neutrality” of hair is pH 6, which is the point of greatest stability of the hair structure. This intrinsic point of neutrality must be restored after permanent waving to maintain the structural integrity of the hair. Closely related to hair fiber water hydrophilicity is its thermal behavior. Dry hair can withstand temperatures during heat styling for a short time when it is dry; it is recommended not to exceed 200 °C on virgin hair, and 180 °C on chemically treated hair. In the wet state, however, hair can suffer structural damage at much lower temperatures, sometimes even below 100 °C. Hair fibers undergo longitudinal aging when exposed to external factors such as grooming or environmental influences [2, 5, 6]. The hair emerges at the scalp surface in a virgin state but can suffer structural damage due to mechanical effects (e.g. friction during styling), temperature changes (e.g. heat styling), or UV‐radiation. Free radicals can further induce structural damage at the surface and the cortex of the hair fiber via chemical oxidation. The result of this range of insults is chemical and mechanical wear of the hair. Some of the chemical changes are cleavage of the hair’s proteins, conversion of amino acids, oxidation of cystine to cysteic acid, and decomposition of histidine. As a consequence, stability and elasticity of the hair, especially at its ends, decreases, flexibility is reduced, and hair fibers get more rigid and brittle, a process termed “hair weathering.” Unsightly split ends often occur and the hair tends to feel dry (Figure 33.3). The structural differences between hair roots and hair ends require special attention in hair permanent waving because the ends of hair – due to their structural and chemical changes – react faster to the perming process and tend to break more easily (Figure 33.4). The protein content of a normal hair at ambient conditions is approximately 80 wt% [2, 7]. Further components are approximately 5 wt% internal lipids; <1 wt% trace elements and metals; 14 wt% water. Of the 22 amino acid types found in hair, the most important is “L‐cystine” (56‐89‐3), a sulfur‐containing amino acid (Figure 33.5), which facilitates covalent cross‐linking between two different protein chains. Up to the high amount of 9 mol% (750 μmol/g hair) [8] is typical for cornified tissues, such as hair, nail, hooves, horn, or cornea. Because of the covalent cross‐links between amino acids of different proteins, hair demonstrates high mechanical strength and shear resistance, insolubility in water but is also prone to swelling. Figure 33.3 The worn cuticle‐free split end of hair (SEM × 800). Figure 33.4 Coiled (alfa‐helical) and amorphous molecules of hair proteins are cross‐linked by disulfide bonds inside the cortical cells. Helical proteins are stabilized by hydrogen bonds. Figure 33.5 Chemical formula of the amino acid “cystine.” Two types of proteins constitute the hair content: low and high sulfurous proteins. It is their typical arrangement that differentiates hair proteins from proteins in the rest of the body: Figure 33.4 shows the network of hair proteins, with the “disulfide bonds” marked in yellow, which gives hair properties similar to a fiber‐reinforced plastic [9] or the elastic rubber of a tire. This special architecture is the source of hair’s elasticity. Perming agents cleave disulfide bonds in these matrix proteins, causing the plasticization that is necessary for shaping hair. However, an excess of a reducing agent is necessary to achieve this, as cross‐linked sulfur proteins in the cuticle make the hair mantle hard to dissolve and penetrate. Additional support of the chemical network is provided by the acid and basic amino acids in hair protein which make up more than 40% of the hair’s substance. These form low‐energy bridges, salt linkages, and hydrogen bonds, which are also cleaved and reformed during the permanent waving process. Because of hair’s great elasticity and strong resilient forces, it quickly resumes its original straight shape after being re‐shaped without structural changes. Therefore, it has to be softened and subsequently re‐hardened chemically to maintain a conformation change. Especially with permanent waving, it is important to select a reversible reaction to allow repeated treatments without hair destruction. The sulfur bridges of the amino acid cystine, linking the proteins, are best suited [5, 10, 11]. The conditions for permanent waving to be well tolerated are A permanent wave occurs with two solutions: Figure 33.6 Chemical reaction formulas 0–2. It is crucial to keep in mind that permanent waving is a two‐step procedure with the chemical reaction and physical effects running in parallel to achieve sufficient results (Figures 33.7 and 33.8) [12–14]: reduction of disulfide‐bonds, softening of hair, lateral swelling and length contraction, stress development and protein flow, then re‐oxidation of cystine bonds and de‐swelling, and fixation of the new curly shape. Table 33.1 summarizes how hair reacts chemically and physically during each of the permanent waving steps. Usually, only 85% of the cleaved disulfide is reformed during neutralization. Some hair cystine oxidizes to give cysteic acid (Figure 33.6, formula 2), which renders hair more hydrophilic, incompletely cross‐linked, and more vulnerable to subsequent treatments. Therefore, permed hair gradually loses its curl and relaxes to a straight hair conformation again (for additional details, the reader is referred to Robbins [2] and Wickett and Savaides [15]).
CHAPTER 33
Permanent Hair Waving
Introduction
Hair physiology
Permanent wave hair relevant properties
Hair geometry
Hair and water interaction
Hair aging
Hair chemical structure
Chemophysical principles of hair waving
Perm products and types
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree