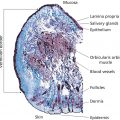
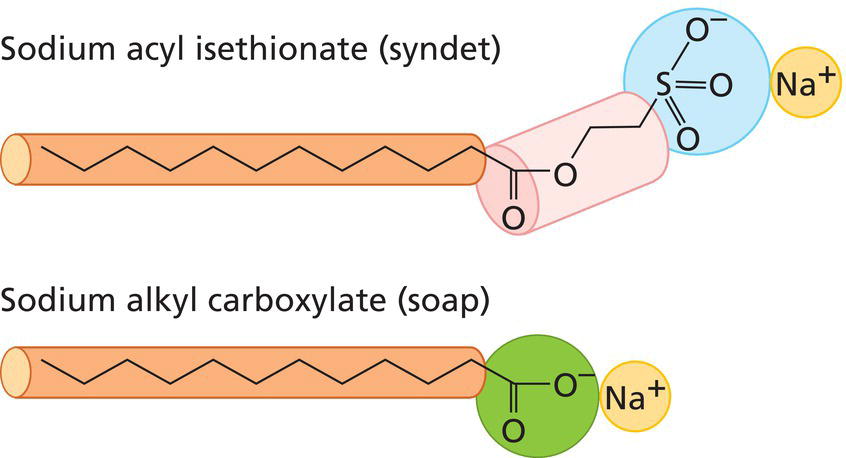
K.P. Ananthapadmanabhan and Stacy Hawkins Unilever HPC R&D, Trumbull, CT, USA Anecdotally, soap was discovered by prehistoric man, noticing a waxy residue in the ashes of an evening campfire around a burnt piece of animal carcass. The waxy material was soap. Potash from the ashes (KOH) had hydrolyzed triglyceride from animal fat to produce potassium soap and glycerol. Actual historical records show soap‐like materials in use by Sumerians in 2500 BC and there are references to soap in Greek and Roman records and by the Celts in northern Europe. As European civilizations emerged from the Dark Ages in the 9th and 10th centuries soap making was well‐established and centered in Marseilles (France), Savona (Italy), and Castilla (Spain). In those days, soap was a luxury affordable only by the very rich. Mass manufacture of soap started in the 19th century and was well established by the turn of the century with individually wrapped and branded bars. Synthetic detergents emerged in the 20th century, primarily for fabric washing products. While there are many types of synthetic detergent, very few are suitable for making cleansing bars. It is difficult to make a solid product that is able to retain a solid form during multiple encounters with water and at the same time able to resist cracking, crumbling, and hardening when drying between uses. Soap is ideal for making bars but that is not to say that some of the early soap bars did not dry out and develop cracks or become soft and mushy in humid environments. Modern manufacturers are able to formulate soap bars to control the physical behavior in use and when drying between uses. Despite conversion of many bar users to liquid format, bars remain an important format in the United States and even more so globally. Figure 12.1 shows the segmentation of the US Personal Wash market with all bar cleansers holding 37% share of value. While soap‐based bars continue to dominate in total volume, the higher value of synthetic detergent bars command 70% of the dollar share. The wide range of soap bars available in the skin marketplace today might suggest a wide range of functionality but this is not the case. To develop new claims and gain shelf space in big supermarkets, manufacturers create variants by minor modifications of their basic bar types – the functional properties of soap bar variants are usually very similar – they all lather and they all clean. Cleansing bars are made of surfactants that are solid at room temperature and readily soluble in water. While there are scores of commercially available surfactants only two, alkyl carboxylate (soap) and acyl isethionate (syndet), are used on a large scale for manufacture of cleansing bars (Figure 12.2). These two surfactant types are quite different, leading to different sensory experiences for the consumer and also differences in their interactions with skin. Soap and syndet have in common that they have the physical properties required to be processed into bars that can withstand the challenges of use in the home. As bars they must have a consistent performance – they must lather easily when new but just as readily as the bar is used up over a period of weeks or months. They should produce lather quickly and easily and should not feel gritty in use. The rate of wear should be optimum, neither too fast nor too slow. They must dry quickly after use but must not crack; they should not break apart if dropped, and should not absorb water and become mushy in a humid environment, like a bathroom. There are not many surfactants that can satisfy this list of seemingly simple practical requirements. Broadly speaking, there are two types of manufacturing processes for making cleansing bars: (1) a continuous process of milling, extrusion, and stamping; and (2) a batch process of melt casting. Figure 12.1 Dollar segmentation of the US cleanser market shows that bars continue to command significant share. Bars in the US alone have a total value of $1.6 billion of which syndet bars are the majority share (2013 data). Figure 12.2 Schematic representation of the molecular structures of soap (sodium alkyl carboxylate) and syndet (sodium acyl isethionate) showing the difference in head group structure and size. The continuous process starts with synthesis of the basic surfactant, alkyl carboxylate, and then processing this as a solid through various steps during which other ingredients are added until the final composition is attained. After milling and mixing steps to ensure homogeneity, the compounded soap is extruded as a continuous bar which is chopped and stamped into the individual bar shape of the final product. The technical demands of the continuous process impose constraints on composition and ingredient addition – but it is the fastest and cheapest way to make a cleansing bar. The essence of the melt cast approach is to make the surfactant and add any desired ingredients to form a hot liquid melt which is poured into individual bar size casts and allowed to set as it cools. This is a much more expensive process but allows for a wider range of additional ingredients in the product formulation. The continuous process is used for most of the mass market bars and the melt cast process for specialist bars often sold in boutiques, custom outlets, and department stores. There are several major compositional types of soap bar with distinct bar properties and in use behaviors – speed and type of lather, rate of use up, aroma, skin compatibility, tendency for mush, etc. Most bars are either basic or superfatted soap. Basic soaps are blends of medium chain length fatty acid sodium salts (Figure 12.2). Superfatted soaps are similar but with additional fatty acid. There are other categories of soap bars based on the use of specialist ingredients: transparent bars, antibacterial bars, and deodorant bars. There are large numbers of specialist bars that are simply soap containing a wide range of colors, fragrances, and emotive ingredients such as vitamins, aloe, chamomile, and other natural extracts. The emotive ingredients in specialist bars are there to appeal to the senses and emotions with no real expectation that they have any detectable benefit for the skin. Soap is the sodium salt of a fatty acid. As the salts of weak acids, soaps form alkaline solutions as they dissociate in water. The pH of soap is typically in the pH range 9–11. This is not sufficient to be overtly irritating to skin but is sufficiently high to negatively impact the pH‐dependent processes of the stratum corneum which has a natural pH of around 5.5. The fatty acids used in soap making are natural, derived from animal or plant sources, with the most common chain lengths in the range C12 (e.g. coconut fatty acid) to C18 (e.g. tallow/rendered animal fat). C12–14 soaps are soluble and lather easily. C16–18 soaps are less soluble but good for forming solid bars. The plant oils used in soap making are mostly triglycerides and when treated with lye and/or caustic soda they hydrolyze to the fatty acid sodium salts (soap) and glycerol. Simple soaps are good cleansers but also drying to skin. Less drying soaps are made by adjusting the soap‐making process to leave an excess of free fatty acid in the final soap composition (superfatted soaps). This excess fatty acid reduces the lipid stripping and drying effects of a soap bar to a small extent. Beauty soaps are typically superfatted soaps. There are several types of transparent or semi‐transparent soap bars. The earliest was a rosin glycerin soap bar developed by Andrew Pears in 1789. The ingredients of Pears patented transparent soap were sodium palmitate, natural rosin, glycerine, water, C12 soap, rosemary extract, thyme extract, and fragrance. The Pears soap of today is made by essentially the same process, which involves dissolving the raw soap and other ingredients in alcohol, pouring into molds followed by up to 3 months of evaporation and drying. A different type of transparent bar was introduced in 1955 by Neutrogena based on a patented formulation invented by a Belgian cosmetic chemist, Edmond Fromont. His novel formulation was based on triethanolamine soap (in other words, soap where the neutralizing cation is triethanolamine instead of the usual sodium). The ingredients of the Neutrogena bar are triethanolamine stearate, C12–18 soaps, glycerine, water, and a range of minor ingredients including a little lanolin derivative and fragrance. Triethanolamine forms acid soaps so the pH of the Neutrogena bar at pH 8–9 is lower than a regular soap with sodium as the cation. Medicated or antibacterial soaps are a large subcategory of the bar soap market. These products are basic soaps containing one of a limited number of approved antibacterial agents. Some of these products are positioned as deodorant soap to inhibit the odor‐producing bacteria of the axilla. However, some are positioned for germ inhibition which has come under increased scrutiny. Washing with any soap is effective for removing and killing the bacteria on skin and the contribution of added antibacterial agents is controversial. Because soap is cheap and easy to manufacture the cleansing bar market has remained predominantly soap bars. However, there has been one nonsoap bar technology that has achieved a significant place in the US market over the last 60 years and is now extending its reach to other regions of the world. This product, introduced to the US market in 1957 as the Dove bar, is based on patented acyl isethionate as the surfactant component in combination with stearic acid which has a dual function of providing the physical characteristics for forming a stable bar and also acting as a significant skin protecting and moisturizing ingredient. The high level of stearic acid in the Dove bar is the basis of the one‐quarter moisturizing cream in the product. When the patents for this novel technology ran out, several other acyl isethionate bars were introduced in the US market including Caress, Olay, Cetaphil, and Aveeno. Interestingly, this mild nonsoap technology pioneered in bar with acyl isethionate has only recently been translated in 2009 as a mild and moisturizing surfactant component in liquid cleanser formats. Similar to what was observed in the approach to developing ultra‐mild bars, it is expected that this liquid acyl isethionate technology will spread widely where liquid cleansers comprise a large portion of market share, thus reaffirming the value of this “bar technology” for cleansing. It is of interest that soap bars and syndet bars are self‐preserved in the sense that they provide a hostile environment for microorganisms and do not need to contain a preservative to maintain product quality. Washing with soap removes dirt and grime from skin and is very effective for removing germs and preventing the spread of infection. There is an appreciation that some soaps are harsh and others mild, but washing with soap is so routine and commonplace that most people give no thought to the cleansing process or its impact on skin. This is a mistake. Research over the last few years has revealed several mechanisms by which soap interacts with skin structures to adversely affect normal functioning. It is now clear that mild cleansing has significant benefits for both diseased and healthy skin. Mild cleansing can help reduce the dry skin symptoms of common skin conditions such as eczema, acne, and rosacea simply by eliminating of the aggravations of harsh soap. Mild cleansing can also enhance the attractiveness of normal skin by allowing it to better retain moisture. As described in other chapters of this book, the outer layer of skin, the stratum corneum, is a very effective barrier to the penetration of microorganisms and chemicals unless compromised by damage, disease, or a intrinsic weakness caused by one of the genetic variations now known to impact the functioning of the stratum corneum. Whatever the normal state of the stratum corneum for an individual, the most challenging (i.e. potentially damaging) environmental factor, apart from industrial exposure to solvents and other harsh chemicals, is cleansing. And yet cleansing is a key element of good everyday skincare and there is much variation in the damaging potential of different cleansing products including cleansing bars. Understanding how cleansing products impact skin and knowing the mildest cleansing product technologies is a basic requirement for achieving fundamental skin care. The properties of soap that make it an effective cleanser also determine that it can be drying and irritating to skin. The high charge density of the carboxyl head group of the soap molecule promotes strong protein binding which is bad for skin. Soap binds strongly to stratum corneum proteins and disturbs the water‐holding mechanisms of the corneocytes. Soaps also denature stratum corneum enzymes essential for corneocytes maturation and desquamation. The result is an accumulation of corneocytes at the skin surface and the characteristic scaly, flaky, roughness associated with dry skin. In addition to damaging proteins, soap and other cleansers can disrupt and strip out the lipid bi‐layers of the stratum corneum. The bipolar structure of the soap molecule is similar to the bipolar structure of the three major lipid types that make up the lipid bi‐layers of the stratum corneum (fatty acids, cholesterol, and ceramides). Soap disrupts the bi‐layer structure of these lipids in the stratum corneum and thereby reduces the effectiveness of the stratum corneum water barrier. Transepidermal water loss (TEWL) is increased through the leaky barrier. Also, disruption of the structured lipid matrix around stratum corneum cells (corneocytes) allows the highly soluble components of the skin’s natural moisturizing factor (NMF), contained in the protein matrix of the corneocytes, to leach out. Leaching is increased by further cleansing or even simply by contact with water. This process explains the paradox that water is often a major factor for causing dry skin. Effects on the key lipid structures of the stratum corneum add to the damage caused by soap–protein interactions and exacerbate the development of skin dryness – remembering that dry skin is not simply a lack of moisture but a disturbance of normal stratum corneum function with retention and accumulation of superficial corneocytes. The build‐up of corneocytes at the skin surface is responsible for many symptoms associated with “dry” skin – scaling, flaking, roughness, dull appearance (due to light scattering), tightness, loss of resilience/flexibility/elasticity, and ultimately cracking and irritation. All soaps have the ability to induce dry and irritated skin and these effects are most evident in challenging environmental condition – cold or hot temperatures with low humidity, excessive exposure to solar UV radiation, and prolonged exposure to wind. The drying potential of soap varies according to composition such as the balance between soluble (C12–14) and less soluble chain lengths (C16–18) of the fatty acids most commonly used to make soap – the higher the soluble component the more drying the soap. Superfatted soaps are a little milder than simple soaps, and triethanolamine soap and glycerol bars the mildest of the commonly available soap bars. Synthetic detergent bars (syndet bars) have been available on the US market for 50 years and represent a clear technological difference from soap‐based cleansing bars. Nearly all common synthetic detergent bars are based on an anionic surfactant, acyl isethionate. At the time of writing (2015) these bars account for 40% of the cleansing bars sold in the United States. Alkyl glycerol ether sulfonate (AGES) and monoalkyl phosphate (MAPS) are two of a small number of other synthetic detergents that have been tried for manufacture of cleansing bars but none of these have been successful in the US market. Ironically, because syndet bars are shaped like soap bars and used for cleansing just like a soap bar, most people believe that synthetic detergent bars are just another variety of soap. Most consumers are unaware that there is a fundamental compositional difference between soap and syndet bars that impacts their interactions with skin such that syndet bars are milder than soap bars during cleansing. There is a greater difference between soap and syndet cleansing in terms of healthy and attractive skin than most people realize. It is important for healthcare professionals and dermatologists to appreciate the difference between soap and syndet bars because studies show the difference in mildness is very relevant for their patient groups (see studies described below). Soap (alkyl carboxylate) and syndet (acyl isethionate) are both anionic surfactants and like all anionic surfactants, they interact with skin proteins and skin lipids. But because of the difference in head group, physical chemistry soap interactions are more intense leading to a higher potential for inducing dryness and irritation. The carboxylate head group is compact, leading to a high charge density that facilitates binding and denaturing of proteins. By contrast, the isethionate head group is large and diffused, producing a low charge density and less ability to interact with proteins (Figure 12.2). A second important factor contributing to the mildness of the isethionate syndet bar is the ability to formulate acyl isethionate with high levels of stearic acid without losing the ability to lather. In fact, the lather is more dense and creamy than the lather of a typical soap bar. The stearic acid component of the isethionate syndet bar acts as a moisturizing cream and deposits on skin during cleansing, adding to the relative mildness of these types of bar. Stearic acid also enhances the mildness of the surfactant base by modulating the isethionate micellar properties. Superfatting is, in principle, a similar way to reduce the harshness of plain soap but the results are much more modest because the initial harshness of soap is higher than syndet and the upper limit of practical superfatting is closer to 10% compared to the 20–25% fatty acid that can be formulated in an isethionate bar. The stearic acid deposited from the syndet bar during cleansing has been shown from tape strip measurements of deuterated fatty acids to penetrate into deeper layers of the stratum corneum. The importance of such penetration of fatty acids into deeper layers in enhancing the stratum corneum barrier becomes even more significant in the light of some of the recent results reported by Bouwstra and team on the fate of fatty acids delivered to deeper layers of the skin. Specifically, Thakoersing et al. [6] have shown from in‐vitro skin model systems that fatty acids such as stearic acid and palmitic acids penetrating into deeper layers can be utilized by skin as precursors and they can be elongated into longer chain fatty acids that are integral part of the stratum corneum lipids. These observations further warrant more in‐vivo studies to assess the fate of fatty acids deposited from syndet bars and their role in enhancing the skin barrier. Another important difference between soap and syndet bars is pH. Soap has an alkaline pH typically around pH 10–11 whereas isethionate/stearic acid bars are close to pH neutral with a pH of a little over 7. The pH of glycerol bars is in the range pH 8–9. These differences in pH have an effect on the interaction of cleansing bars with the stratum corneum.
CHAPTER 12
Bar Cleansers
Introduction
Cleansing bars – historical perspective
Formulation technology of cleansing bars

Continuous processing
Batch processing
Soap bars
Basic soap
Superfatted soap bars
Transparent soaps
Antibacterial and deodorant soap bars
Nonsoap detergent bars – syndet bars
Preservatives
Impact of cleansing bars on skin structure and function
Surfactant interaction with the skin–stratum corneum
Soap bar interactions with the stratum corneum
Synthetic detergent bar interactions with the stratum corneum
The role of stearic acid
The role of pH
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree